Chapter 4: Q.20. (page 128)
Question: Calculate the destabilization present in each eclipsed conformation.
a.
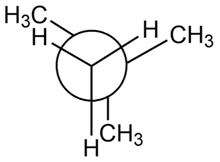
b.
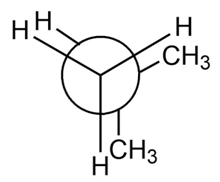
Short Answer
Answer
a. The destabilization energyis 3.8 kcal/mol.
b. The destabilization energy is 4.2 kcal/mol.
Learning Materials
Features
Discover
Chapter 4: Q.20. (page 128)
Question: Calculate the destabilization present in each eclipsed conformation.
a.

b.

Answer
a. The destabilization energyis 3.8 kcal/mol.
b. The destabilization energy is 4.2 kcal/mol.
All the tools & learning materials you need for study success - in one app.
Get started for free
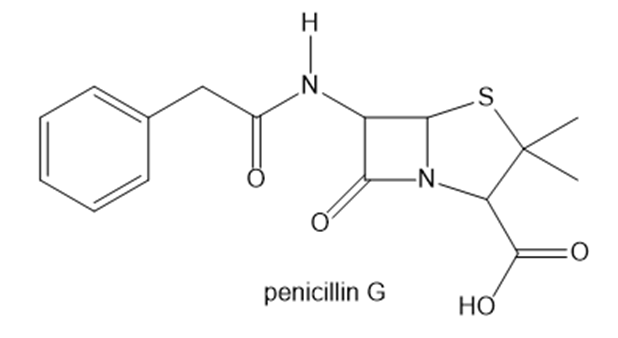
Question: Although penicillin G has two amide functional groups, one is much more reactive than the other. Which amide is more reactive and why?
Question: Name each alkane using the ball-and-stick model, and classify each carbon as , or .
a.
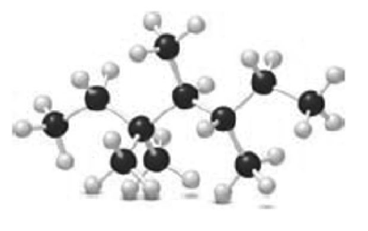
b.
Give the structure corresponding to each IUPAC name.
a. 1,2-dimethylcyclobutane
b. 1,1,2-trimethylcyclopropane
c. 4-ethyl-1,2-dimethylcyclohexane
d. 1-sec-butyl-3-isopropylcyclopentane
e. 1,1,2,3,4-pentamethylcycloheptane
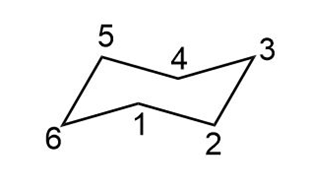
Question: Using the cyclohexane with the C’s numbered as shown, draw a chair form that fits each description.
a. The ring has an axial group at C1 and an equatorial OH on C2.
b. The ring has an equatorial group on C6 and an axial OH group on C4.
c. The ring has equatorial OH groups on C1, C2, and C5.
Question: The torsional energy in propane is 14 kJ/mol (3.4 kcal/mol). Because each H,H eclipsing interaction is worth 4.0 kJ/mol (1.0 kcal/mol) of destabilization, how much is one H, eclipsing interaction worth in destabilization? (See Section 4.10 for an alternate way to arrive at this value.)
What do you think about this solution?
We value your feedback to improve our textbook solutions.