Chapter 4: Q14. (page 146)
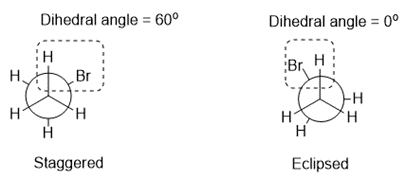
Draw the staggered and eclipsed conformations that result from rotation around the C-C bond in.
Short Answer
The staggered and eclipsed conformations are shown below:
Learning Materials
Features
Discover
Chapter 4: Q14. (page 146)
Draw the staggered and eclipsed conformations that result from rotation around the C-C bond in.
The staggered and eclipsed conformations are shown below:

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Draw the three constitutional isomers having molecular formula that contain a five-membered ring and two methyl groups as substituents. For each constitutional isomer that can have cis and trans isomers, draw the two stereoisomers.
Question: Convert each of the following structures into its more stable chair form. One structure represents menthol and one represents isomenthol. Menthol, the more stable isomer, is used in lip balms and mouthwash. Which structure corresponds to menthol?
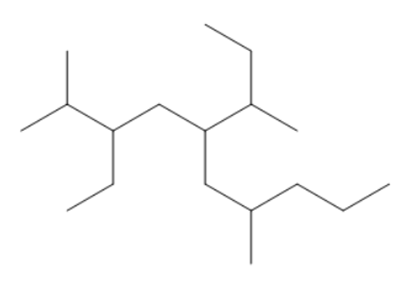
Question:Give IUPAC name of each compound.
a.
b.
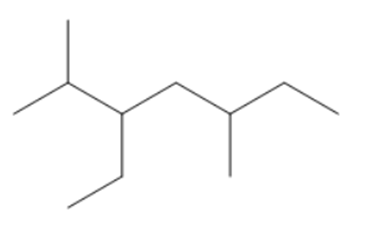
c.
d.
Question: Consider the tricyclic structure B.
(a) Label each substituent on the rings as axial or equatorial.
(b) Draw B using chair conformations for each six-membered ring.
(c) Label the atoms on the ring fusions (the carbons that join each set of two rings together) as cis or trans to each other.
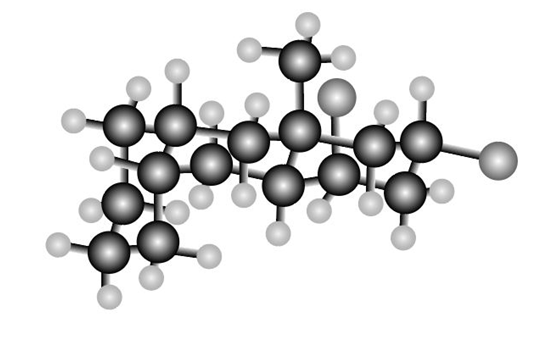
B
Question: Give the IUPAC name for each of the five constitutional isomers of molecular formula in Problem 4.3.
What do you think about this solution?
We value your feedback to improve our textbook solutions.