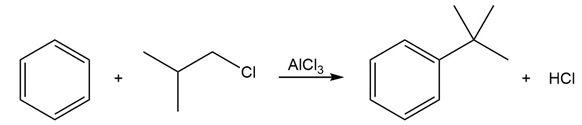
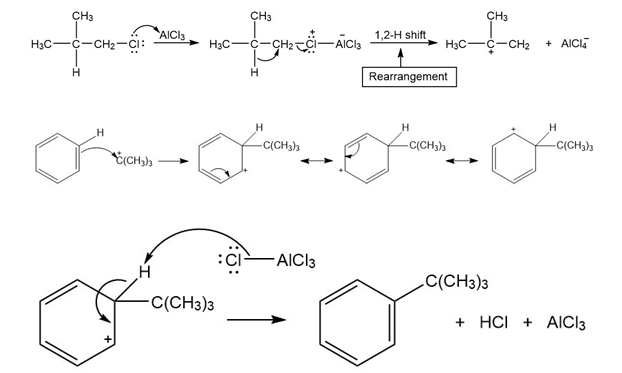
Chapter 18: 8P (page 688)
Draw a stepwise mechanism for the following reaction.
Short Answer
Learning Materials
Features
Discover
Chapter 18: 8P (page 688)
Draw a stepwise mechanism for the following reaction.


All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Ibufenac, a para-disubstituted arene with the structure HO2CCH2C6H4CH2CH(CH3)2, is a much more potent analgesic than aspirin, but it was never sold commercially because it caused liver toxicity in some clinical trials. Devise a synthesis of ibufenac from benzene and organic halides having fewer than five carbons.
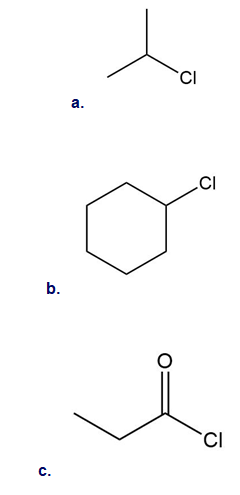
What product is formed when benzene is treated with each organic halide in the presence of .
Question: Draw a stepwise, detailed mechanism for the dienone–phenol rearrangement, a reaction that forms alkyl-substituted phenols from cyclohexadienones.

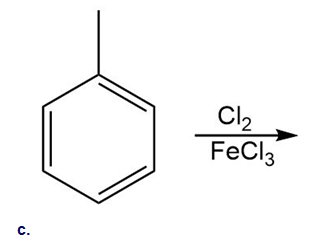
Draw the products of each reaction.
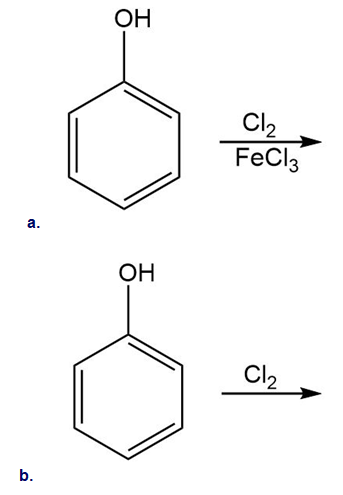
Which substituents have an electron-withdrawing and which have an electron-donating inductive effect
a
b
c
What do you think about this solution?
We value your feedback to improve our textbook solutions.