Chapter 18: 24 P (page 708)
Draw the products of each reaction.
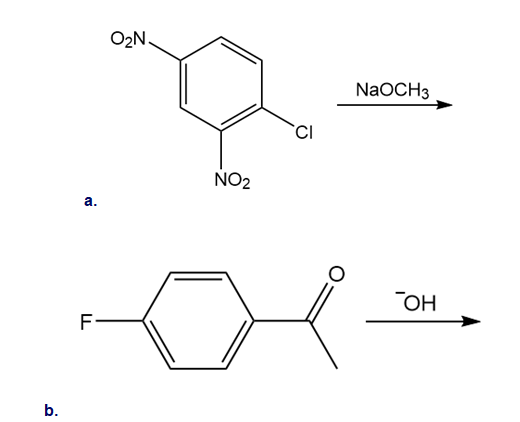
Short Answer
Learning Materials
Features
Discover
Chapter 18: 24 P (page 708)
Draw the products of each reaction.


All the tools & learning materials you need for study success - in one app.
Get started for free
Draw the products of each reaction.
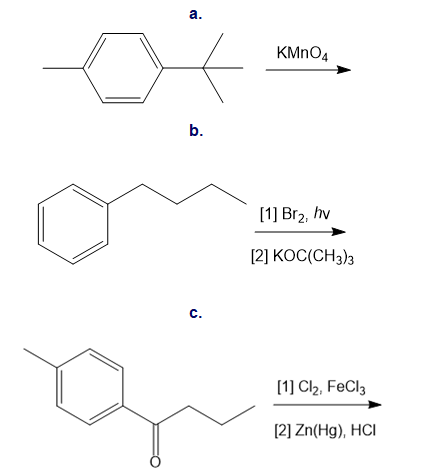
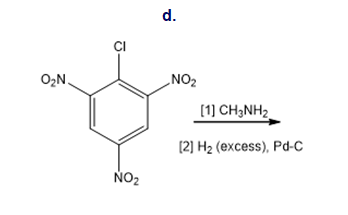
Classify each substituent as electron donating or electron withdrawing.

Question: Using resonance structures, explain why a nitroso group (-NO) is an ortho, para director that deactivates a benzene ring toward electrophilic attack.
Question: Draw a stepwise, detailed mechanism for the dienone–phenol rearrangement, a reaction that forms alkyl-substituted phenols from cyclohexadienones.

Rank the compounds in each group in order of increasing reactivity in electrophilic aromatic substitution: (a) ; (b) .
What do you think about this solution?
We value your feedback to improve our textbook solutions.