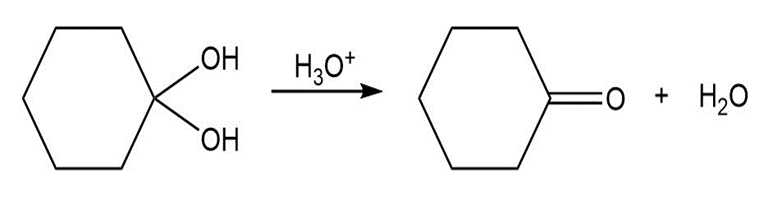
Chapter 21: Aldehydes and Ketones-Nucleophilic Addition
1P
Rank the following compounds in order of increasing reactivity towards nucleophilic attack.
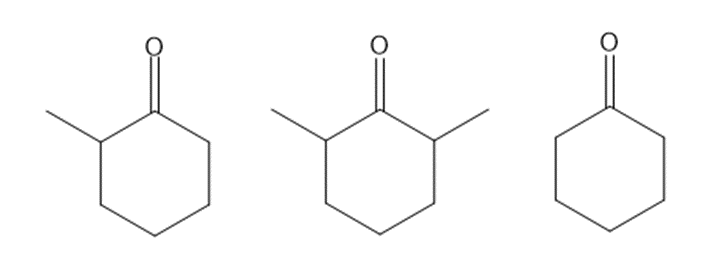
20P
What starting materials are needed to prepare each alkene by a Wittig reaction? When there are two possible routes, indicate which route, if any, is preferred.
a.
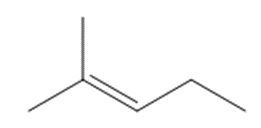
b
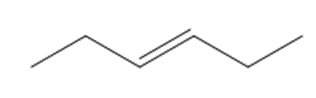
c.
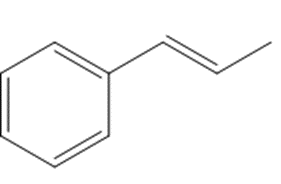
21P
Show two methods to synthesize each alkene: a one-step method using a Wittig reagent, and a two-step method that forms a carbon–carbon bond with an organometallic reagent in one of the steps.
a.
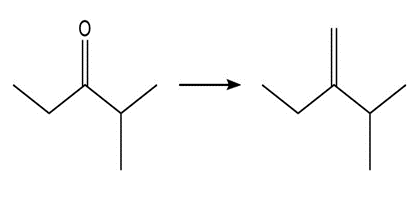
b.
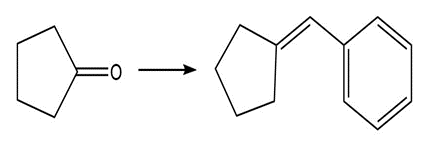
22P
�ٰ����� t��� p���ǻ�ܳ��� f�ǰ������ w���� C��3CH2CH2CH2NH2reacts with each carbonyl compound in the presence of mild acid.
a.
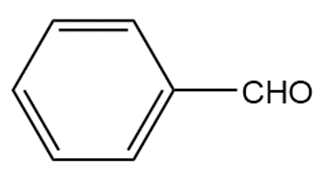
b.
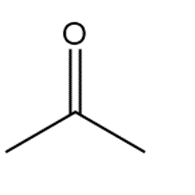
c.
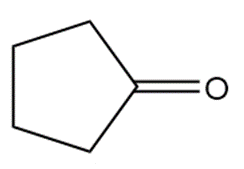
23P
What 1° amine and carbonyl compound are needed to prepare each imine?
a.
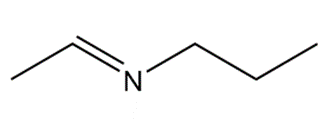
b.
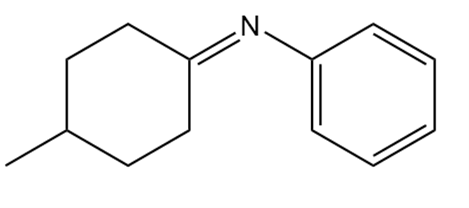
24P
What two enamines are formed when 2-methylcyclohexanone is treated with (CH3)2NH?
25P
What carbonyl compound and amine are formed by the hydrolysis of each compound?
a.
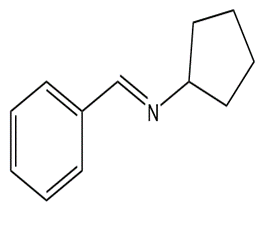
b.
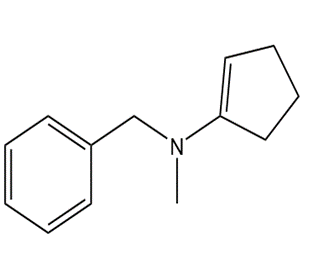
c.
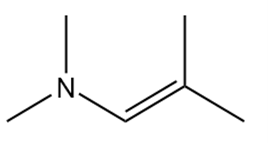
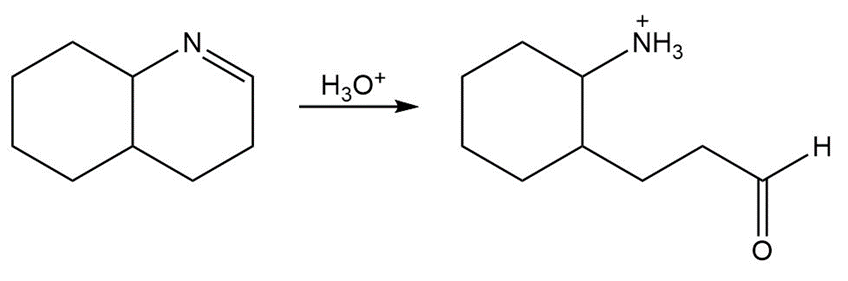
26P
Draw a stepwise mechanism for the following imine hydrolysis
27P
Which compound in each pair forms the higher percentage of gem-diol at equilibrium?
a.
 or
or
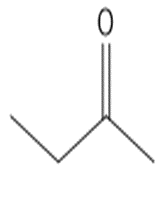
b.
 or
or
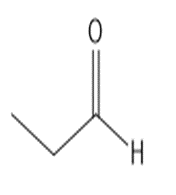
28P
Draw a stepwise mechanism for the following reaction