Chapter 21: 26P (page 817)
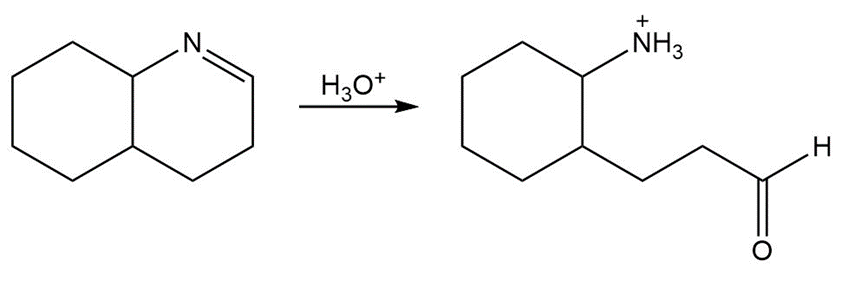
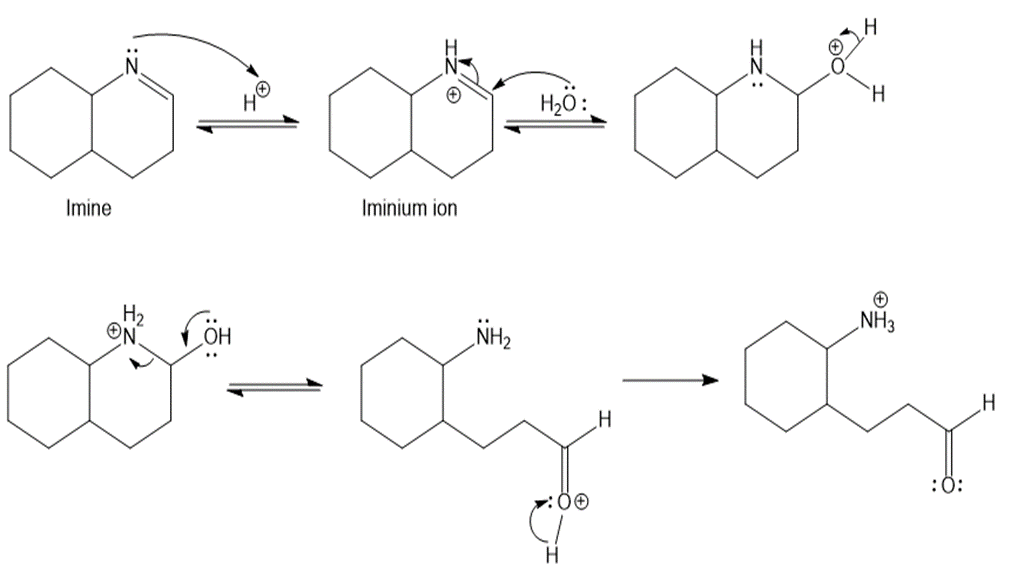
Draw a stepwise mechanism for the following imine hydrolysis
Short Answer
Learning Materials
Features
Discover
Chapter 21: 26P (page 817)
Draw a stepwise mechanism for the following imine hydrolysis


All the tools & learning materials you need for study success - in one app.
Get started for free
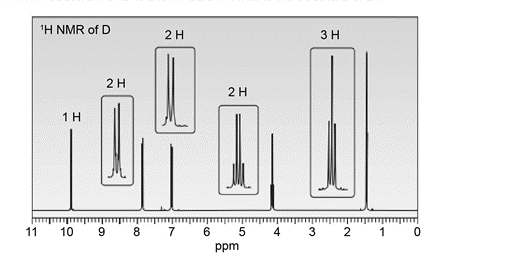
An unknown compound D exhibits a strong absorption in its IR spectrum at 1692 cm-1 . The mass spectrum of D shows a molecular ion at m/z = 150 and a base peak at 121. The 1H- NMR spectrum of D is shown below. What is the structure of D?
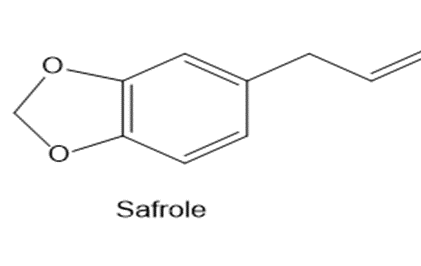
Safrole is a naturally occurring acetal isolated from sassafras plants. Once used as a common food additive in root beer and other beverages, it is now banned because it is carcinogenic. What compounds are formed when safrole is hydrolysed with aqueous acid?
Use the 1 H NMR and IR data to determine the structure of each compound.

Draw the products of the following Wittig reactions
a.
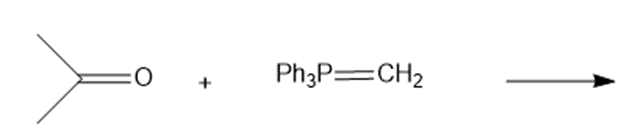
b.
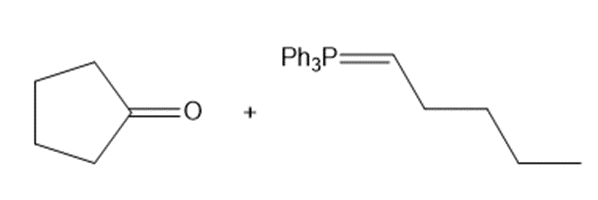
Design a stepwise synthesis to convert cyclopentanone and 4-bromobutanal to hydroxy aldehyde A.

What do you think about this solution?
We value your feedback to improve our textbook solutions.