Chapter 21: 27P (page 817)
Which compound in each pair forms the higher percentage of gem-diol at equilibrium?
a.
 or
or
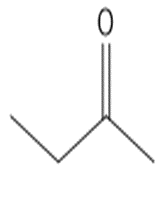
b.
 or
or

Short Answer
a.
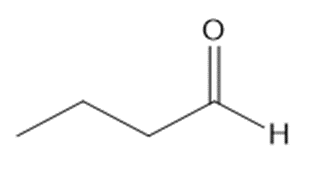
b.

Learning Materials
Features
Discover
Chapter 21: 27P (page 817)
Which compound in each pair forms the higher percentage of gem-diol at equilibrium?
a.
 or
or

b.
 or
or

a.

b.

All the tools & learning materials you need for study success - in one app.
Get started for free
What lactol (cyclic hemiacetal) is formed from intramolecular cyclization of each hydroxy aldehyde?
a.
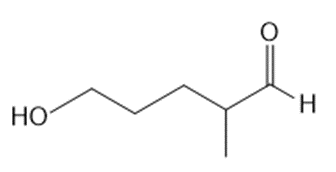
b.

Give the structure corresponding to each name:
(a) sec-butyl ethyl ketone
(b) methyl vinyl ketone
(c) p-ethylacetophenone
(d) 3-benzoyl-2-benzylcyclopentanone
(e) 6,6-dimethylcyclohex-2-enone
(f) 3-ethylhex-5-enal
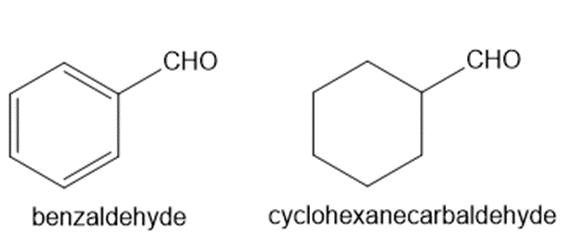
Explain why benzaldehyde is less reactive than cyclohexanecarbaldehyde towards nucleophilic attack.
Draw a stepwise mechanism for the following reaction
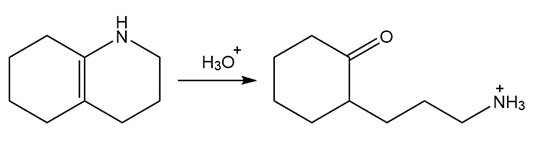
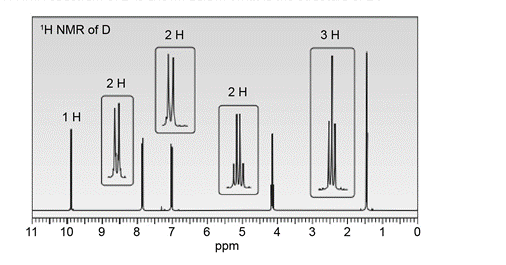
An unknown compound D exhibits a strong absorption in its IR spectrum at 1692 cm-1 . The mass spectrum of D shows a molecular ion at m/z = 150 and a base peak at 121. The 1H- NMR spectrum of D is shown below. What is the structure of D?
What do you think about this solution?
We value your feedback to improve our textbook solutions.