Chapter 21: 36P (page 817)
What lactol (cyclic hemiacetal) is formed from intramolecular cyclization of each hydroxy aldehyde?
a.
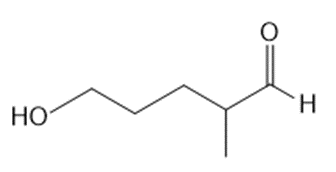
b.
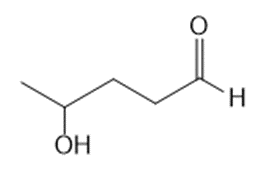
Short Answer
a.
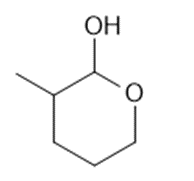
b.
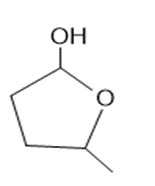
Learning Materials
Features
Discover
Chapter 21: 36P (page 817)
What lactol (cyclic hemiacetal) is formed from intramolecular cyclization of each hydroxy aldehyde?
a.

b.

a.

b.

All the tools & learning materials you need for study success - in one app.
Get started for free
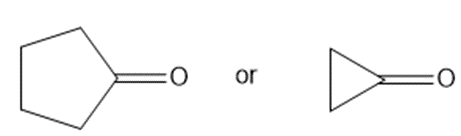
Which carbonyl group in each pair absorbs at a higher frequency?
a.
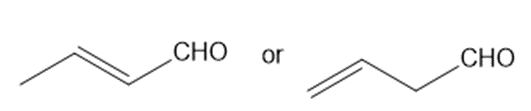
b.
Draw the products of the following Wittig reactions
a.
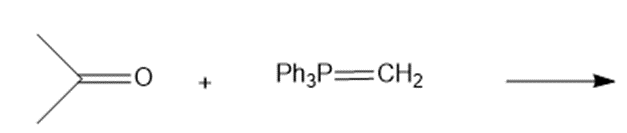
b.
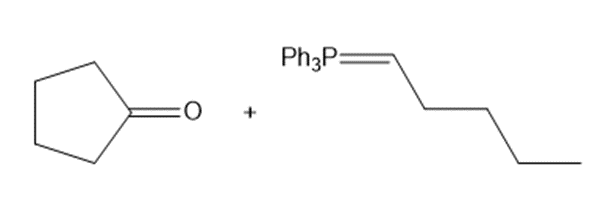
Draw a stepwise mechanism for the following reaction that converts a dicarbonyl compoundto a furan.
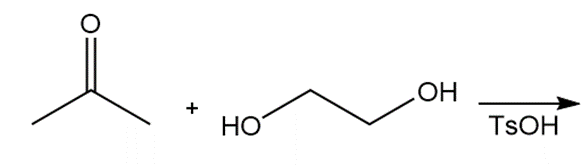
Draw the products of each reaction.
a.
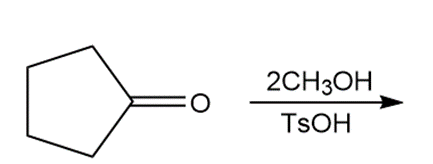
b.
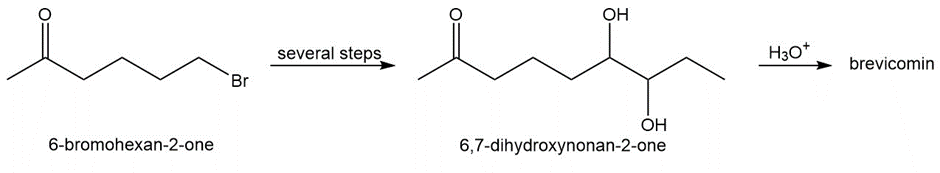
Brevicomin, the aggregation pheromone of the western pine bark beetle, contains a bicyclic bridged ring system and is prepared by the acid-catalyzed cyclization of 6,7-dihydroxynonan-2-one.
a. Suggest a structure for brevicomin.
b. Devise a synthesis of 6,7-dihydroxynonan-2-one from 6-bromohexan-2-one. You may also use three-carbon alcohols and any required organic or inorganic reagents.
What do you think about this solution?
We value your feedback to improve our textbook solutions.