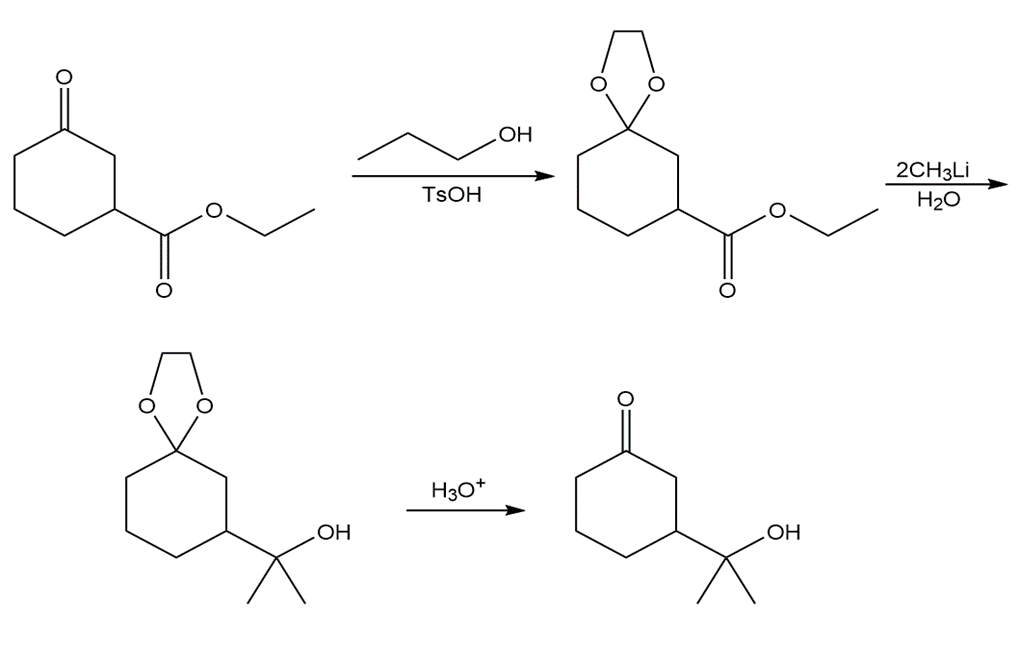
Chapter 21: 35P (page 817)
How would you use a protecting group to carry out the following transformation?
Short Answer
Learning Materials
Features
Discover
Chapter 21: 35P (page 817)
How would you use a protecting group to carry out the following transformation?

All the tools & learning materials you need for study success - in one app.
Get started for free
Rank the following compounds in order of increasing reactivity towards nucleophilic attack.
Draw a stepwise mechanism for the following reaction
Give the structure corresponding to each name:
(a) sec-butyl ethyl ketone
(b) methyl vinyl ketone
(c) p-ethylacetophenone
(d) 3-benzoyl-2-benzylcyclopentanone
(e) 6,6-dimethylcyclohex-2-enone
(f) 3-ethylhex-5-enal
Use the 1 H NMR and IR data to determine the structure of each compound.

What hydrolysis products are formed when the following compound is treated with aqueous acid?
What do you think about this solution?
We value your feedback to improve our textbook solutions.