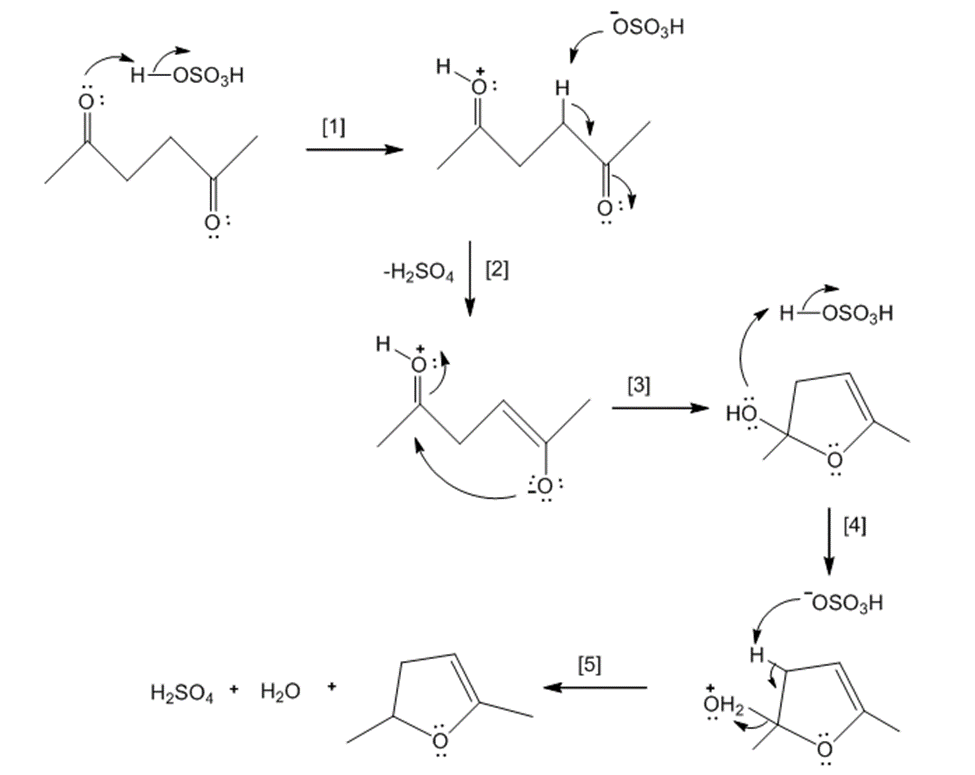
Chapter 21: 68P (page 817)
Draw a stepwise mechanism for the following reaction that converts a dicarbonyl compoundto a furan.
Short Answer
Learning Materials
Features
Discover
Chapter 21: 68P (page 817)
Draw a stepwise mechanism for the following reaction that converts a dicarbonyl compoundto a furan.

All the tools & learning materials you need for study success - in one app.
Get started for free
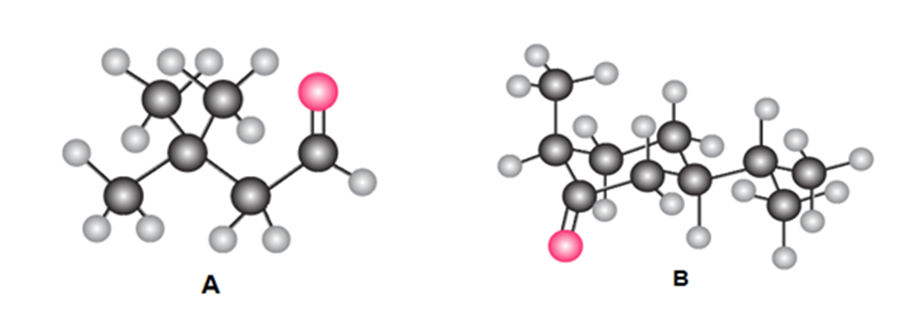
:(a) Give the IUPAC name for A and B. (b) Draw the product formed when A or B is treated with each reagent: [1] NaBH4 [2] CH3MgBr then H2O ; [3] Ph3P=CHOCH3 ;[4] CH3CH2CH2NH2mild acid; [5] HOCH2CH2CH2OH,H+ .
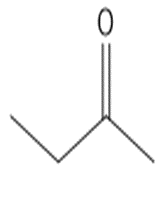
Which compound in each pair forms the higher percentage of gem-diol at equilibrium?
a.
 or
or
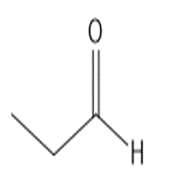
b.
 or
or
What reagents are needed to convert each compound into butanal (CH3CH2CH2CHO )?
a.
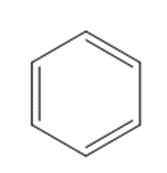
b.
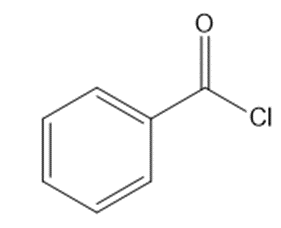
c.
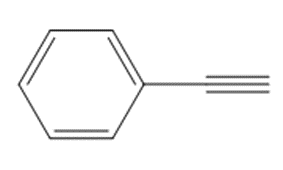
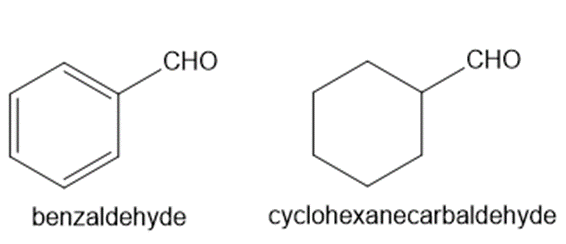
Explain why benzaldehyde is less reactive than cyclohexanecarbaldehyde towards nucleophilic attack.
Treatment of (HOCH2CH2CH2CH2)2CO with acid forms a product of molecular formula C9H16O2and a molecule of water. Draw the structure of the product and explain howit is formed.
What do you think about this solution?
We value your feedback to improve our textbook solutions.