Chapter 21: 28P (page 817)
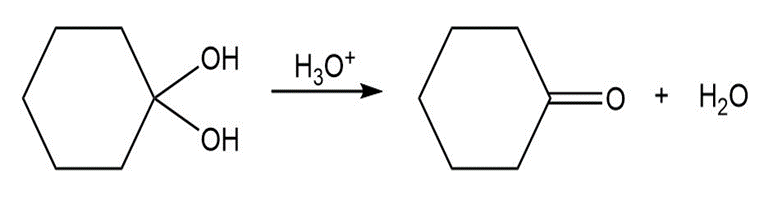
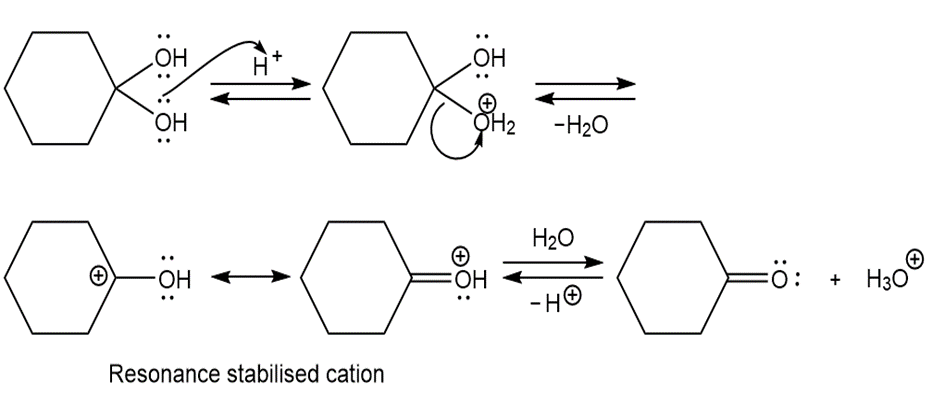
Draw a stepwise mechanism for the following reaction
Short Answer
Learning Materials
Features
Discover
Chapter 21: 28P (page 817)
Draw a stepwise mechanism for the following reaction


All the tools & learning materials you need for study success - in one app.
Get started for free
Hydroxy aldehydes A and B readily cyclize to form hemiacetals. Draw the stereoisomersformed in this reaction from both A and B. Explain why this process gives an opticallyinactive product mixture from A, and an optically active product mixture from B.

What starting materials are needed to prepare each alkene by a Wittig reaction? When there are two possible routes, indicate which route, if any, is preferred.
a.
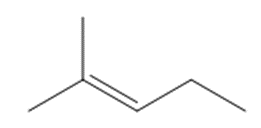
b
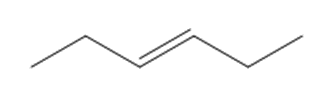
c.
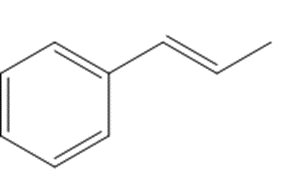
What reagents are needed to convert each compound into butanal (CH3CH2CH2CHO )?
a.
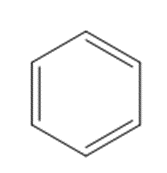
b.
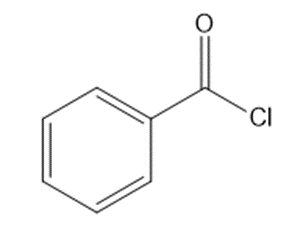
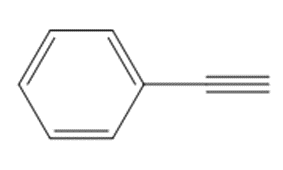
c.
What carbonyl compound and diol are needed to prepare each compound?
a.
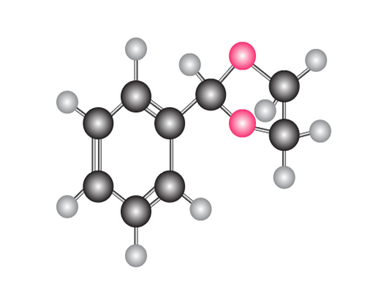
b.
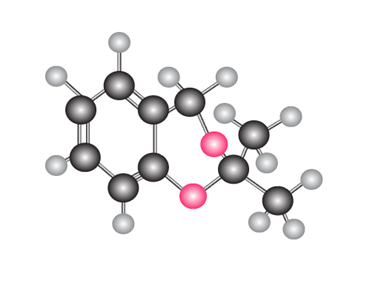
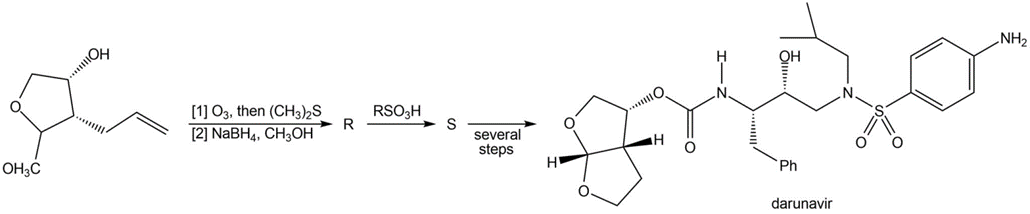
Identify R and S in the following reaction sequence, and draw a mechanism for the conversion of R to S (molecular formula ). S was used in the synthesis of darunavir (trade name Prezista), used to treat HIV.
What do you think about this solution?
We value your feedback to improve our textbook solutions.