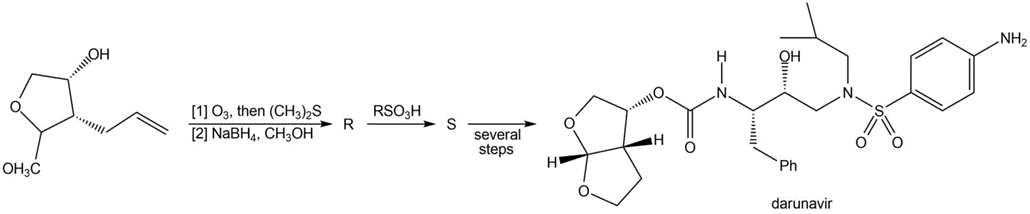
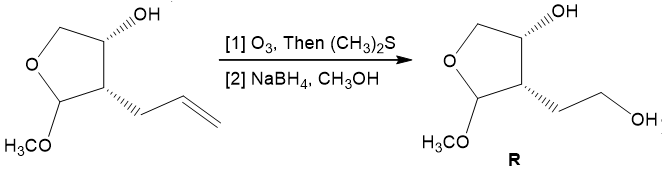
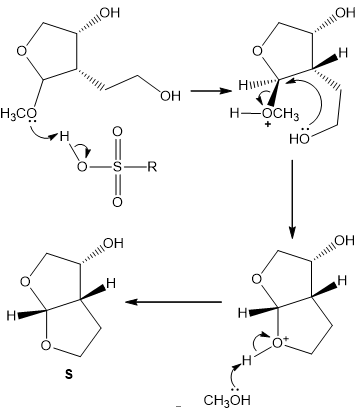
Chapter 21: 85P (page 817)
Identify R and S in the following reaction sequence, and draw a mechanism for the conversion of R to S (molecular formula ). S was used in the synthesis of darunavir (trade name Prezista), used to treat HIV.
Short Answer
Learning Materials
Features
Discover
Chapter 21: 85P (page 817)
Identify R and S in the following reaction sequence, and draw a mechanism for the conversion of R to S (molecular formula ). S was used in the synthesis of darunavir (trade name Prezista), used to treat HIV.



All the tools & learning materials you need for study success - in one app.
Get started for free
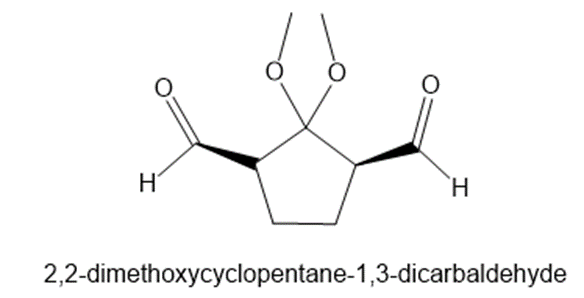
What alkene would yield 2,2-dimethoxycyclopentane-1,3-dicarbaldehyde on treatment withfollowed by ?
Design a stepwise synthesis to convert cyclopentanone and 4-bromobutanal to hydroxy aldehyde A.
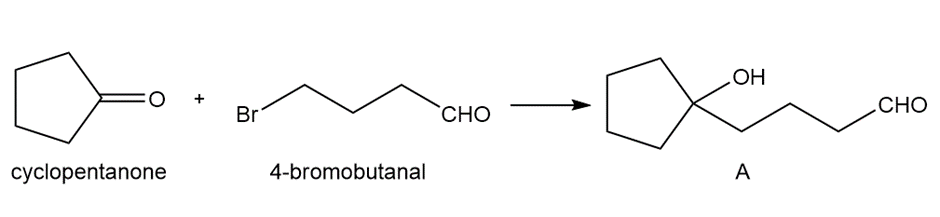
What reagents are needed to convert each compound into butanal (CH3CH2CH2CHO )?
a.
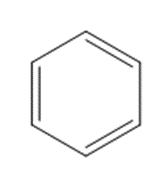
b.
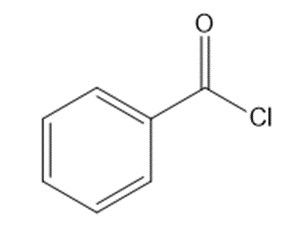
c.
Reaction of 5,5-dimethoxypentan-2-one with methylmagnesium iodide followed by treatment with aqueous acid forms cyclic hemiacetal Y. Draw a stepwise mechanism that illustrates how Y is formed
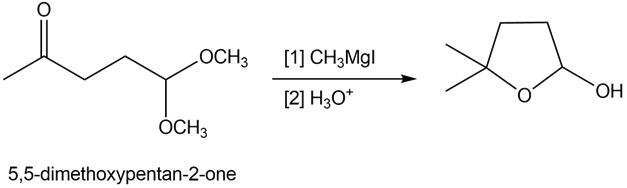
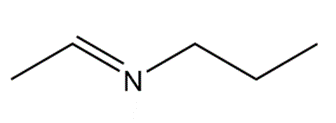
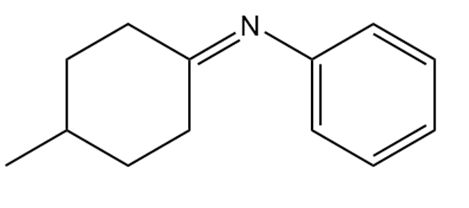
What 1° amine and carbonyl compound are needed to prepare each imine?
a.
b.
What do you think about this solution?
We value your feedback to improve our textbook solutions.