Chapter 21: 86P (page 817)
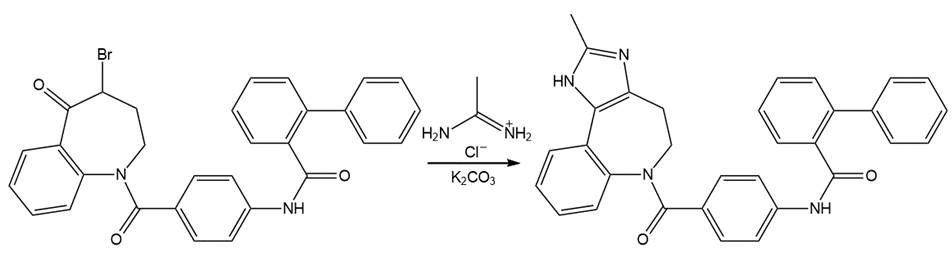
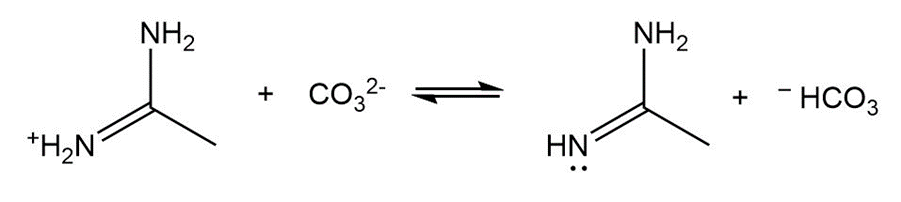
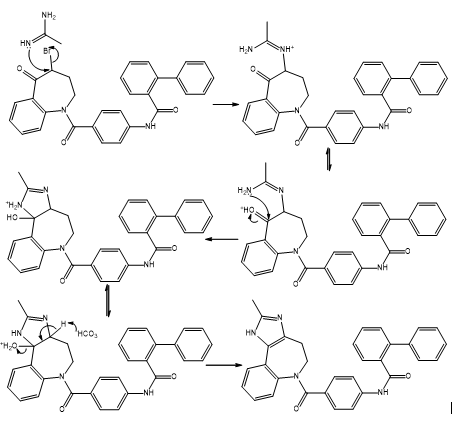
Draw a stepwise mechanism for the following reaction, a key step in the synthesis of conivaptan (trade name Vaprisol), a drug used in the treatment of low sodium levels.
Short Answer
Learning Materials
Features
Discover
Chapter 21: 86P (page 817)
Draw a stepwise mechanism for the following reaction, a key step in the synthesis of conivaptan (trade name Vaprisol), a drug used in the treatment of low sodium levels.



All the tools & learning materials you need for study success - in one app.
Get started for free
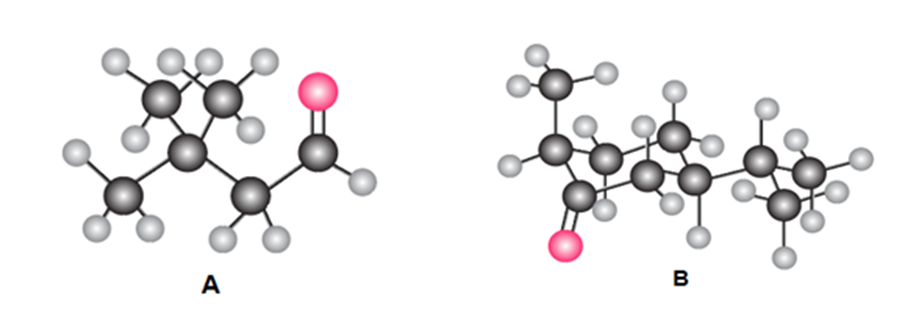
:(a) Give the IUPAC name for A and B. (b) Draw the product formed when A or B is treated with each reagent: [1] NaBH4 [2] CH3MgBr then H2O ; [3] Ph3P=CHOCH3 ;[4] CH3CH2CH2NH2mild acid; [5] HOCH2CH2CH2OH,H+ .
Devise a synthesis of each alkene using a Wittig reaction to form the double bond. You may use benzene and organic alcohols having four or fewer carbons as starting materials and any required reagents
a.
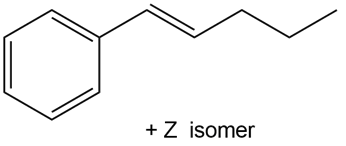
b.
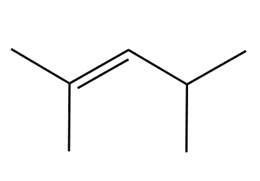
Draw the products of each reaction.
a.
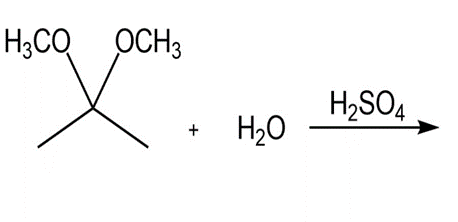
b.
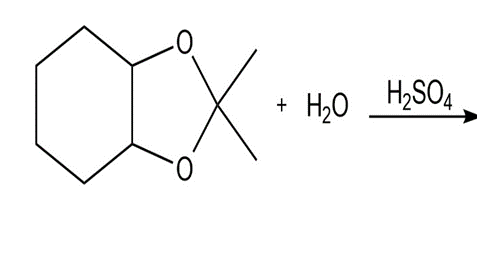
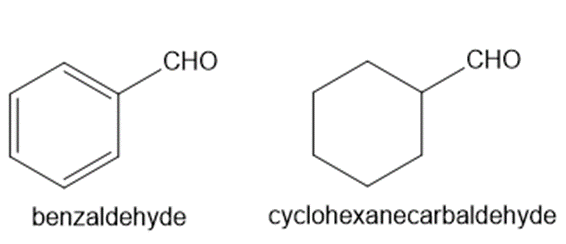
Explain why benzaldehyde is less reactive than cyclohexanecarbaldehyde towards nucleophilic attack.
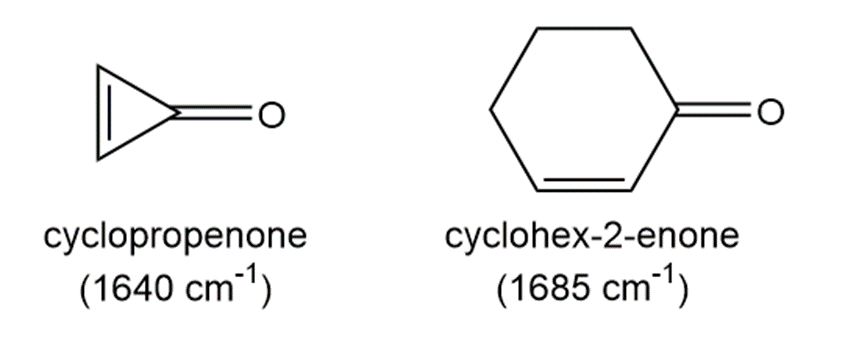
Although the carbonyl absorption of cyclic ketones generally shifts to higher wavenumber with decreasing ring size, C=O of cyclopropenone absorbs at lower wavenumber in its roman IR spectrum than the C=O of cyclohex-2-enone. Explain this observation by using the principles of aromaticity learned in Chapter 17.
What do you think about this solution?
We value your feedback to improve our textbook solutions.