Chapter 21: 23P (page 817)
What 1° amine and carbonyl compound are needed to prepare each imine?
a.
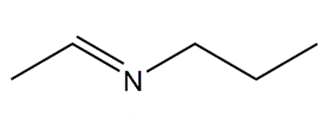
b.
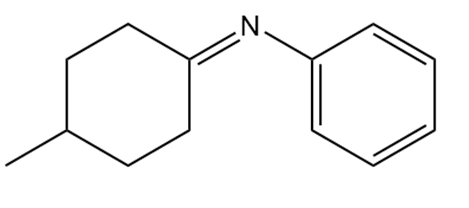
Short Answer
a.
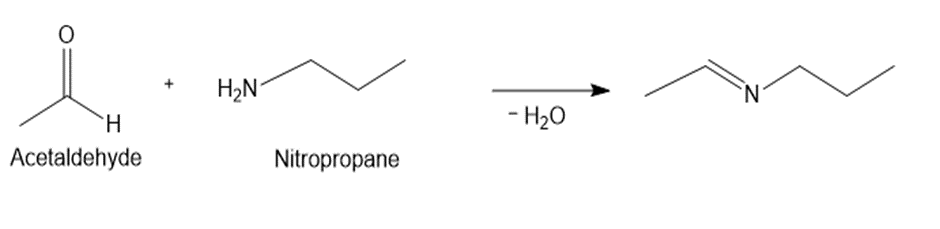
b.
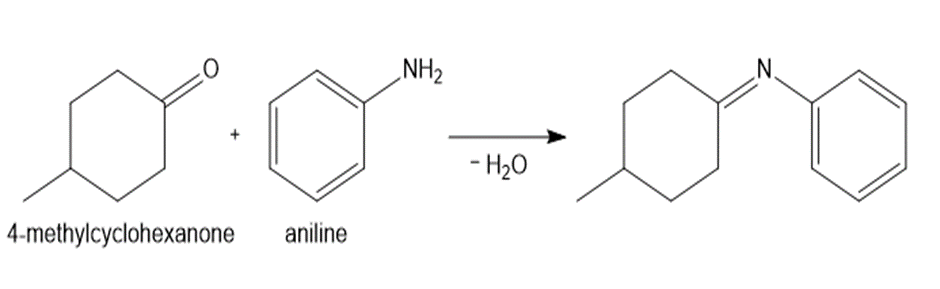
Learning Materials
Features
Discover
Chapter 21: 23P (page 817)
What 1° amine and carbonyl compound are needed to prepare each imine?
a.

b.

a.

b.

All the tools & learning materials you need for study success - in one app.
Get started for free
Show two different methods to carry out the following transformation: a one-step method using a Wittig reagent, and a two-step method using a Grignard reagent. Which route, if any, is preferred?

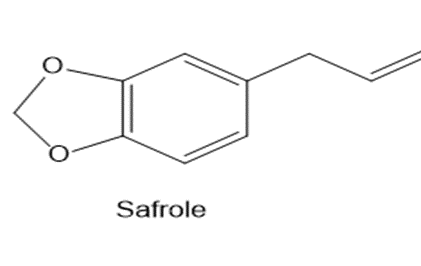
Safrole is a naturally occurring acetal isolated from sassafras plants. Once used as a common food additive in root beer and other beverages, it is now banned because it is carcinogenic. What compounds are formed when safrole is hydrolysed with aqueous acid?
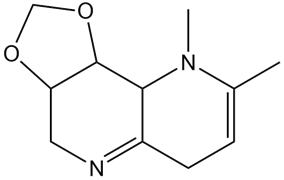
What hydrolysis products are formed when the following compound is treated with aqueous acid?
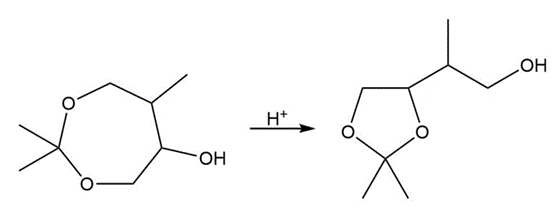
Draw a stepwise mechanism for the following reaction
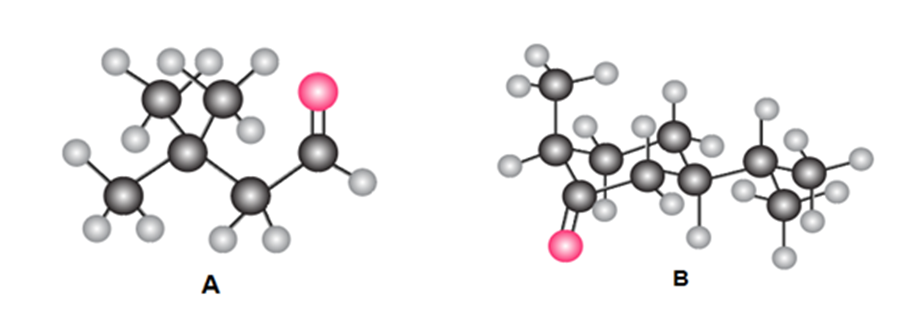
:(a) Give the IUPAC name for A and B. (b) Draw the product formed when A or B is treated with each reagent: [1] NaBH4 [2] CH3MgBr then H2O ; [3] Ph3P=CHOCH3 ;[4] CH3CH2CH2NH2mild acid; [5] HOCH2CH2CH2OH,H+ .
What do you think about this solution?
We value your feedback to improve our textbook solutions.