Maltose is a carbohydrate present in malt, the liquid obtained from barley and other grains. Although maltose has numerous functional groups, its reactions are explained by the same principles we have already encountered.
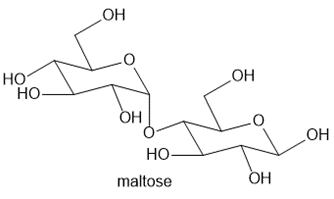
a. Label the acetal and hemiacetal carbons.b. What products are formed when maltose is treated with each of the following reagents: [1] ; [2] and HCl; [3] excess NaH, then excess ?c. Draw the products formed when the compound formed in Reaction [3] of part (b) is treated with aqueous acid.The reactions in parts (b) and (c) are used to determine structural features of carbohydrates like maltose. We will learn much more about maltose and similar carbohydrates in Chapter 28.












