Chapter 21: 22P (page 817)
�ٰ����� t��� p���ǻ�ܳ��� f�ǰ������ w���� C��3CH2CH2CH2NH2reacts with each carbonyl compound in the presence of mild acid.
a.
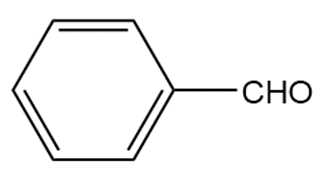
b.
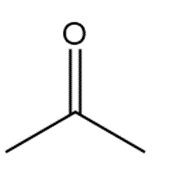
c.
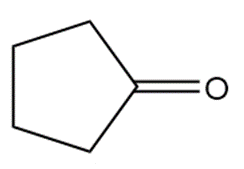
Short Answer
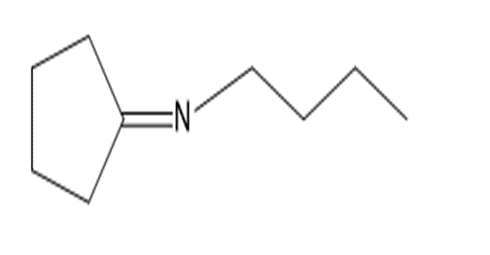
a.
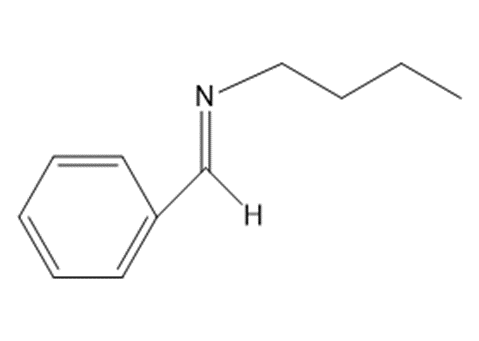
b.
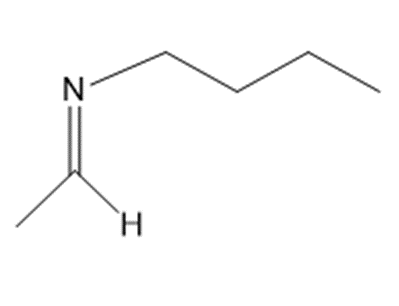
c
Learning Materials
Features
Discover
Chapter 21: 22P (page 817)
�ٰ����� t��� p���ǻ�ܳ��� f�ǰ������ w���� C��3CH2CH2CH2NH2reacts with each carbonyl compound in the presence of mild acid.
a.

b.

c.

a.

b.

c

All the tools & learning materials you need for study success - in one app.
Get started for free
Draw a stepwise mechanism for each reaction.
a.
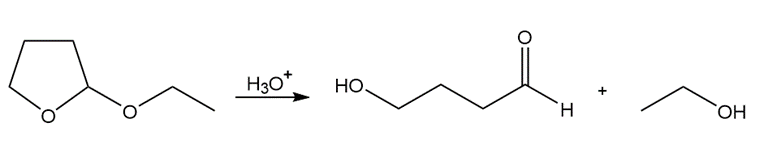
b.
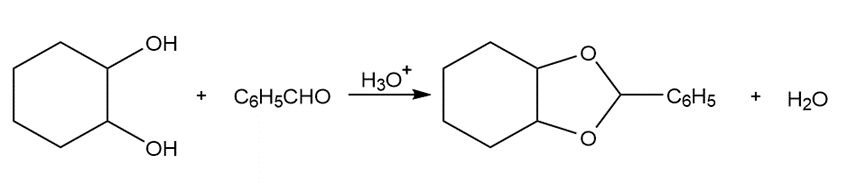
Draw a stepwise mechanism for the following reaction

The boiling point of butan-2-one ( 79.64oC ) is significantly higher than the boiling point of diethyl ether ( 34.6OC), even though both compounds exhibit dipole–dipole interactions and have comparable molecular weights. Offer an explanation.
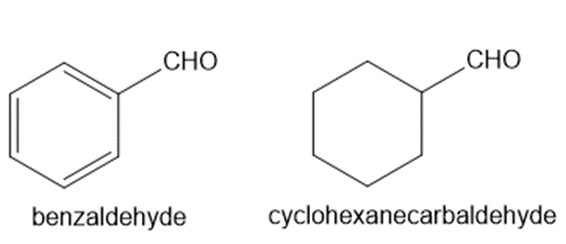
Explain why benzaldehyde is less reactive than cyclohexanecarbaldehyde towards nucleophilic attack.
Use the 1 H NMR and IR data to determine the structure of each compound.

What do you think about this solution?
We value your feedback to improve our textbook solutions.