Chapter 21: 64P (page 817)
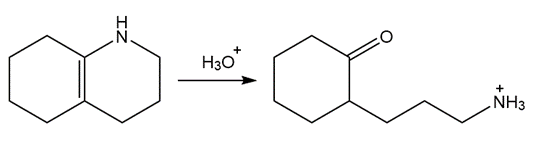
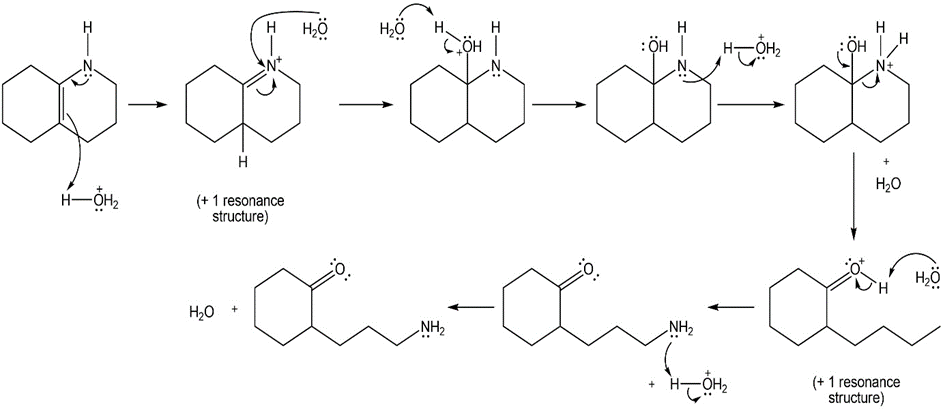
Draw a stepwise mechanism for the following reaction
Short Answer
Learning Materials
Features
Discover
Chapter 21: 64P (page 817)
Draw a stepwise mechanism for the following reaction


All the tools & learning materials you need for study success - in one app.
Get started for free
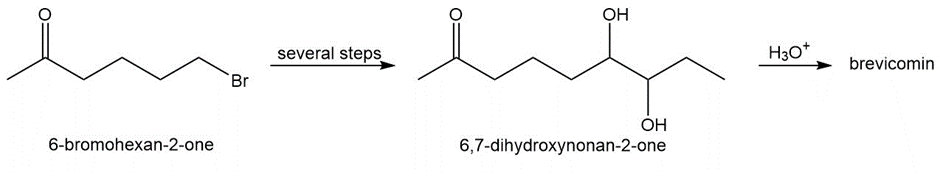
Brevicomin, the aggregation pheromone of the western pine bark beetle, contains a bicyclic bridged ring system and is prepared by the acid-catalyzed cyclization of 6,7-dihydroxynonan-2-one.
a. Suggest a structure for brevicomin.
b. Devise a synthesis of 6,7-dihydroxynonan-2-one from 6-bromohexan-2-one. You may also use three-carbon alcohols and any required organic or inorganic reagents.
�ٰ����� t��� p���ǻ�ܳ��� f�ǰ������ w���� C��3CH2CH2CH2NH2reacts with each carbonyl compound in the presence of mild acid.
a.
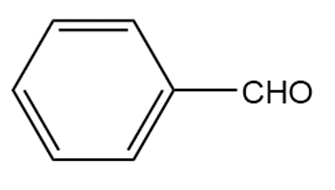
b.
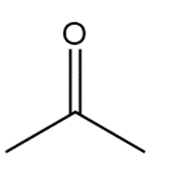
c.
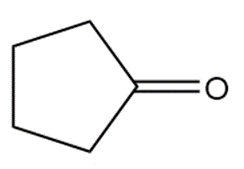
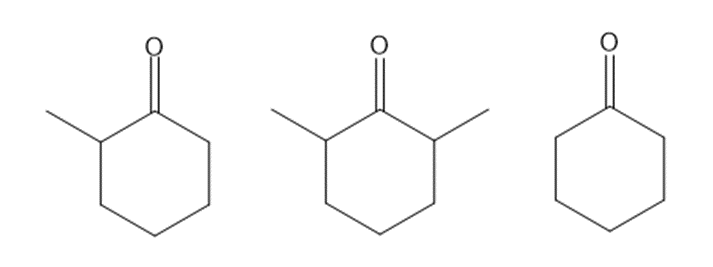
Rank the following compounds in order of increasing reactivity towards nucleophilic attack.
Consider the para-substituted aromatic ketones, NO2C6H4COCH2 (p-nitroacetophenone) and CH3OC6H4COCH3 (p-methoxyacetophenone).
a. Which carbonyl compound is more stable?
b. Which compound forms the higher percentage of hydrate at equilibrium?
c. Which compound exhibits a carbonyl absorption at a higher wavenumber in its IR spectrum?
Explain your reasoning in each part.
Besides the tert-butyldimethylsilyl ethers introduced in Chapter 20, there are many other widely used alcohol protecting groups. For example, an alcohol such as cyclohexanol can be converted to a methoxy methyl ether (a MOM protecting group) by treatment with base and chloromethyl methyl ether, . The protecting group can be removed by treatment with aqueous acid.
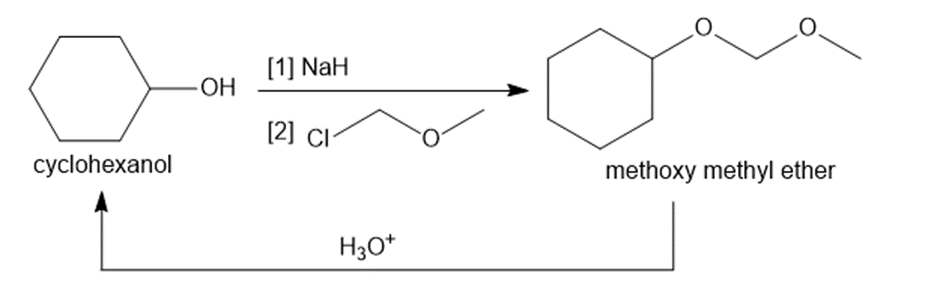
a. Write a stepwise mechanism for the formation of a MOM ether from cyclohexanol.
b. What functional group comprises a MOM ether?
c. Besides cyclohexanol, what other products are formed by aqueous hydrolysis of the MOM ether? Draw a stepwise mechanism that accounts for formation of each product.
What do you think about this solution?
We value your feedback to improve our textbook solutions.