Chapter 21: 65P (page 817)
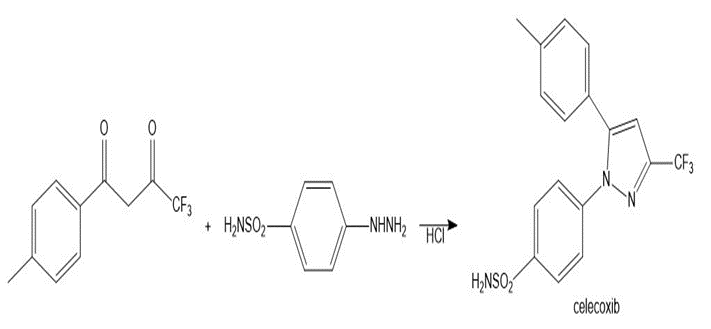
Draw a stepwise mechanism for the following reaction, a key step in the synthesis of the anti-inflammatory drug celecoxib (trade name Celebrex).
Short Answer
Learning Materials
Features
Discover
Chapter 21: 65P (page 817)
Draw a stepwise mechanism for the following reaction, a key step in the synthesis of the anti-inflammatory drug celecoxib (trade name Celebrex).


All the tools & learning materials you need for study success - in one app.
Get started for free
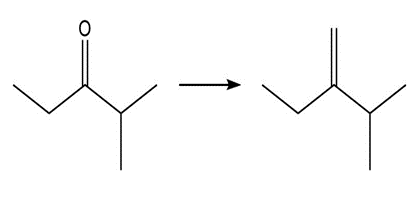
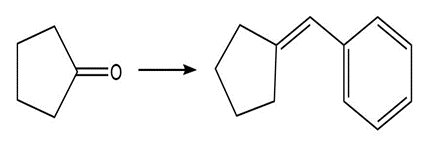
Show two methods to synthesize each alkene: a one-step method using a Wittig reagent, and a two-step method that forms a carbon–carbon bond with an organometallic reagent in one of the steps.
a.
b.
Reaction of 5,5-dimethoxypentan-2-one with methylmagnesium iodide followed by treatment with aqueous acid forms cyclic hemiacetal Y. Draw a stepwise mechanism that illustrates how Y is formed
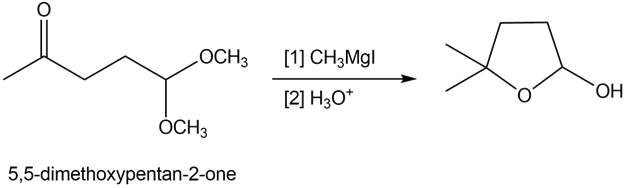
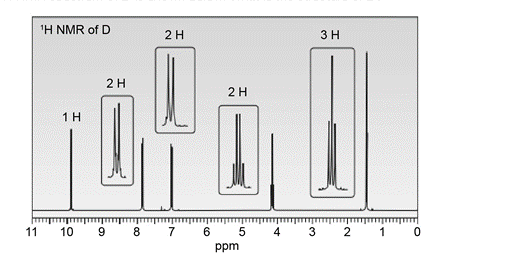
An unknown compound D exhibits a strong absorption in its IR spectrum at 1692 cm-1 . The mass spectrum of D shows a molecular ion at m/z = 150 and a base peak at 121. The 1H- NMR spectrum of D is shown below. What is the structure of D?
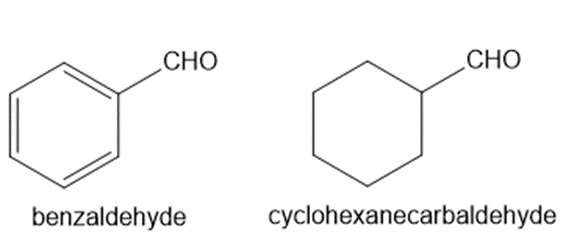
Explain why benzaldehyde is less reactive than cyclohexanecarbaldehyde towards nucleophilic attack.
Draw a stepwise mechanism for the following reaction that converts a dicarbonyl compoundto a furan.
What do you think about this solution?
We value your feedback to improve our textbook solutions.