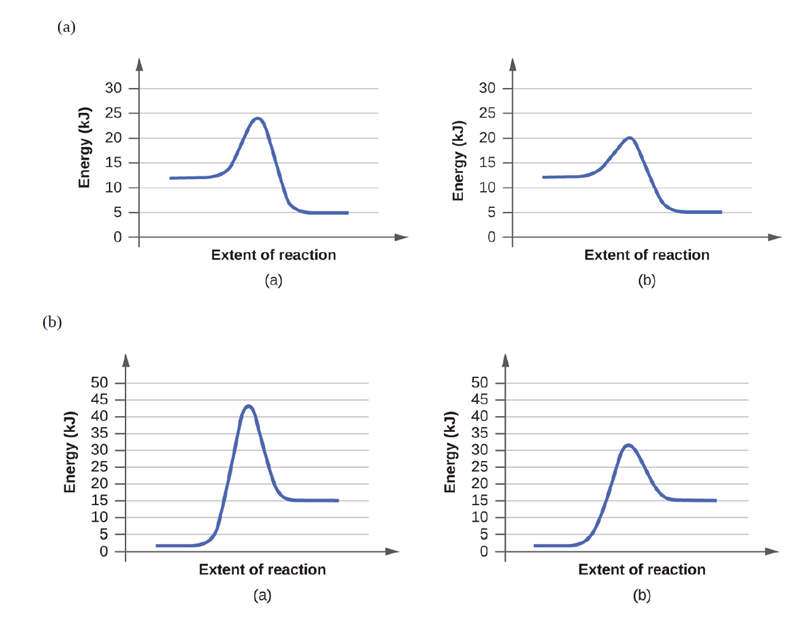
Chapter 12: Q67 E (page 710)
Use the PhET Reactions & Rates interactive simulation (http://openstaxcollege.org/l/ 16PHETreaction) to simulate a system. On the “Single collision” tab of the simulation applet, enable the “Energy view” by clicking the “+” icon. Select the first A + BC⟶AB + C reaction (A is yellow, B is purple, and C is navy blue). Using the “angled shot” option, try launching the A atom with varying angles, but with more Total energy than the transition state. Whathappenswhen the A atom hitstheBC molecule from different directions? Why?
Short Answer
In both cases, the reaction rate depends on the orientation and energy of the reactants.
In all the cases where reactant A hits the molecule BC from different directions, the reaction rate depends on the orientation and energy of the reactants.