Chapter 12: Q5E (page 702)
A study of the rate of the reaction represented as 2A⟶ B gave the following data:
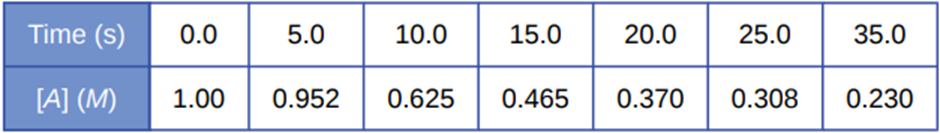
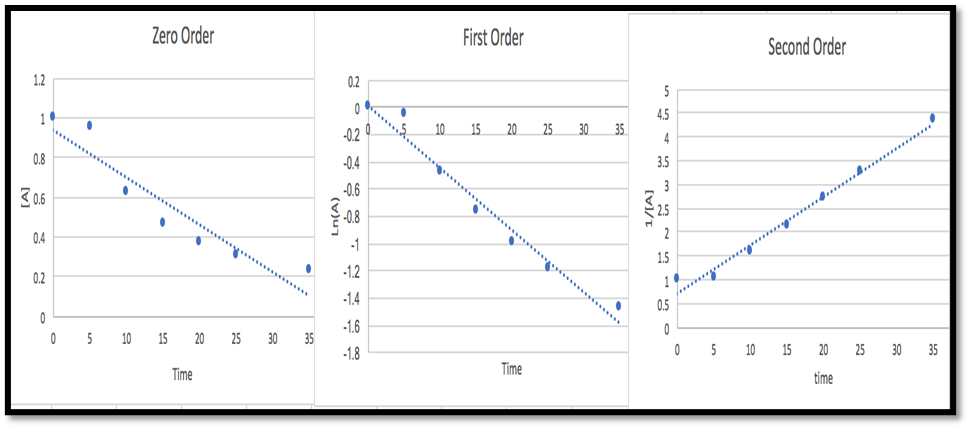
- Determine the average rate of disappearance of A between 0.0 s and 10.0 s, and between 10.0 s and 20.0 s.
- Estimate the instantaneous rate of disappearance of A at 15.0 s from a graph of time versus (A). What are the units of this rate?
- Use the rates found in parts (a) and (b) to determine the average rate of formation of B between 0.00 s and 10.0 s, and the instantaneous rate of formation of B at 15.0 s.
Short Answer
1.The average rate of disappearance of A between 0.0 s and 10.0 s is 0.0374 M/s and Average rate of disappearance of A between 10.0 s and 20.0 s is 0.0255M/s.
2.The instantaneous rate of disappearance of A at 15.0 s from a graph of time versus (A) is 0.05M/s and the units of this rate is \({\bf{mol \times }}{{\bf{L}}^{{\bf{ - 1}}}}{\bf{ \times }}{{\bf{s}}^{{\bf{ - 1}}}}\).
3.The average rate of formation of B between 0.00 s and 10.0 s is get 0.0188M/s and the instantaneous rate of formation of B at 15.0 s is 0.025.