Chapter 2: Atoms, Molecules, and Ions
Q10E
In what way are isotopes of a given element always different? In what way(s) are they always the same?
Q11E
Write the symbol for each of the following ions: (a) the ion with a \(1 + \) charge, atomic number \(55,\) and mass number \(133\)(b) the ion with \(54\) electrons, \(53\) protons, and \(74\) neutrons (c) the ion with atomic number\(15,\) mass number \(31,\) and a \(3 - \) charge (d) the ion with \(24\) electrons, \(30\) neutrons, and a \(3 + \)charge
Q13E
Open theBuild an Atom simulation (http://openstaxcollege.org/l/16PhetAtomBld)and click on the
Atom icon.
a. Pick any one of the first 10 elements that you would like to build and state its symbol.
b. Drag protons, neutrons, and electrons onto the atom template to make an atom of your element.
State the numbers of protons, neutrons, and electrons in your atom, as well as the net charge and mass number.
c. Click on “Net Charge” and “Mass Number,” check your answers to (b), and correct, if needed.
d. Predict whether your atom will be stable or unstable. State your reasoning.
e. Check the “Stable/Unstable” box. Was your answer to (d) correct? If not, first predict what you can do to make a Stable atom of your element, and then do it and see if it works. Explain your reasoning.
Q14E
Open theBuild an Atom simulation (http://openstaxcollege.org/l/16PhetAtomBld)(a) Drag protons, neutrons, and electrons onto the atom template to make a neutral atom of Oxygen-16 and give theisotope symbol for this atom.(b) Now add two more electrons to make an ion and give the symbol for the ion you have created.
Q16E
Determine the number of protons, neutrons, and electrons in the following isotopes that are used in medical diagnoses:
(a) atomic number 9, mass number 18, charge of 1-
b) atomic number 43, mass number 99, charge of 7+
(c) atomic number 53, atomic mass number 131, charge of 1-
(d) atomic number 81, atomic mass number 201, charge of 1+
(e) Name the elements in parts (a), (b), (c), and (d)
Q18E
Give the number of protons, electrons, and neutrons in neutral atoms of each of the following isotopes:
\(\begin{aligned}{}\begin{aligned}{{}{}}{\left( a \right){\rm{ }}{}_5^{10}B}\\{\left( b \right){\rm{ }}{}_{80}^{199}Hg}\\{\left( c \right){\rm{ }}{}_{29}^{63}Cu}\\{\left( d \right){\rm{ }}{}_6^{13}C}\end{aligned}\\\left( e \right){\rm{ }}{}_{34}^{77}Se\end{aligned}\)
Q19E
Give the number of protons, electrons, and neutrons in neutral atoms of each of the following isotopes:\(\begin{aligned}{}\left( a \right){\rm{ }}{}_3^7Li{\rm{ }}\\\left( b \right){\rm{ }}{}_{52}^{125}Te{\rm{ }}\\\left( c \right){\rm{ }}{}_{47}^{109}Ag{\rm{ }}\\\left( d \right){\rm{ }}{}_7^{15}N{\rm{ }}\\\left( e \right){\rm{ }}{}_{15}^{31}P\end{aligned}\)
Q1 E
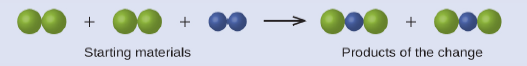
In the following drawing, the green spheres represent atoms of a certain element. The purple spheres represent atoms of another element. If the spheres of different elements touch, they are part of a single unit of a compound. The following chemical change represented by these spheres may violate one of the ideas of Dalton’s atomic theory. Which one?
Q20E
Click on the site (http://openstaxcollege.org/l/16PhetAtomMass) and select the “Mix Isotopes” tab, hide the “Percent Composition” and “Average Atomic Mass” boxes, and then select the element boron.
(a) Write the symbols of the isotopes of boron that are shown as naturally occurring in significant amounts.
(b) Predict the relative amounts (percentages) of these boron isotopes found in nature. Explain the reasoning behind your choice.
(c) Add isotopes to the black box to make a mixture that matches your prediction in (b). You may drag isotopes fromtheir bins or click on “More” and then move the sliders to the appropriate amounts.
(d) Reveal the “Percent Composition” and “Average Atomic Mass” boxes. How well does your mixture match withyour prediction? If necessary, adjust the isotope amounts to match your prediction.
(e) Select “Nature’s” mix of isotopes and compare it to your prediction. How well does your prediction comparewith the naturally occurring mixture? Explain. If necessary, adjust your amounts to make them match “Nature’s”amounts as closely as possible.
Q2.1CYL
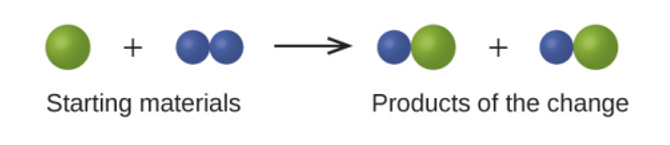
In the following drawing, the green spheres represent atoms of a certain element. The purple spheres represent atoms of another element. If the spheres touch, they are part of a single unit of a compound. Does the following chemical change represented by these symbols violate any of the ideas of Dalton’s atomic Theory? If so, which one?