Chapter 12: Q43 E (page 707)
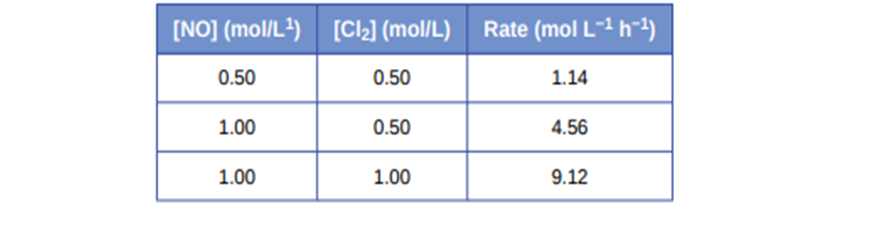
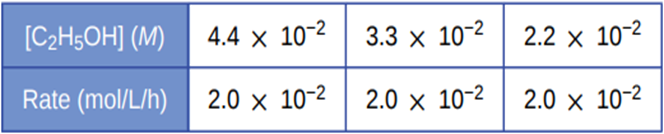
Some bacteria are resistant to the antibiotic penicillin because they produce penicillinase, an enzyme with a molecular weight of \({\bf{3 \times 1}}{{\bf{0}}^{\bf{4}}}\)g/mole that converts penicillin into inactive molecules. Although the kinetics of enzyme-catalysed reactions can be complex, at low concentrations this reaction can be described by a rate equation that is first order in the catalyst (penicillinase) and that also involves the concentration of penicillin. From the following data: 1.0 L of a solution containing 0.15 µg (\({\bf{0}}{\bf{.15 \times 1}}{{\bf{0}}^{{\bf{ - 6}}}}\)g) of penicillinase, determine the order of the reaction with respect to penicillin and the value of the rate constant.
(Penicillin) (M) | Rate (mole/L/min) |
\({\bf{2}}{\bf{.0 \times 1}}{{\bf{0}}^{{\bf{ - 6}}}}\) \(\) | \({\bf{1}}{\bf{.0 \times 1}}{{\bf{0}}^{{\bf{ - 10}}}}\) |
\({\bf{3}}{\bf{.0 \times 1}}{{\bf{0}}^{{\bf{ - 6}}}}\) | \({\bf{1}}{\bf{.5 \times 1}}{{\bf{0}}^{{\bf{ - 10}}}}\) |
\({\bf{4}}{\bf{.0 \times 1}}{{\bf{0}}^{{\bf{ - 6}}}}\) | \({\bf{2}}{\bf{.0 \times 1}}{{\bf{0}}^{{\bf{ - 10}}}}\) |
Short Answer
The order of the reaction with respect to penicillin is first order reaction which can be determined by the use of hit and trial method and the rate constant of the reaction is \({\bf{5}}{\bf{.0 \times 1}}{{\bf{0}}^{{\bf{ - 5}}}}{\bf{L/mole/min}}\).