Chapter 12: Q25 E (page 704)
Nitrogen monoxide reacts with chlorine according to the equation:
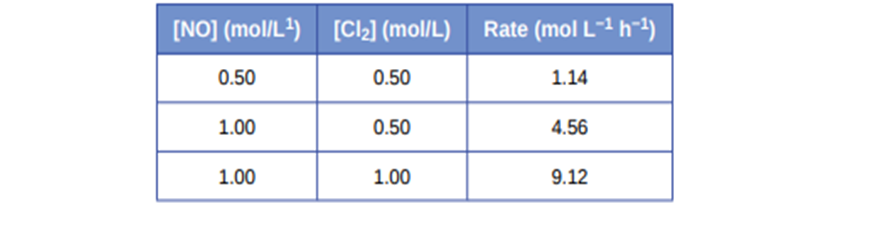
2NO(g) + Cl\({}_2\)(g)⟶ 2NOCl(g) The following initial rates of reaction have been observed for certain reactant concentrations:
What is the rate law that describes the rate’s dependence on the concentrations of NO and Cl2? What is the rate constant? What are the orders with respect to each reactant?
Short Answer
Rate law that describes the rate’s dependence on the concentration of NO and Cl is
represented as
\({\bf{rate = k(NO}}{{\bf{)}}^{\bf{2}}}{{\bf{(Cl)}}^{\bf{1}}}\)
The value of the rate constant is\({\bf{9}}{\bf{.1}}\,{{\bf{L}}^{\bf{2}}}\,{\bf{mo}}{{\bf{l}}^{{\bf{ - 2}}\,}}{{\bf{h}}^{{\bf{ - 1}}}}\)
The order with respect to NO is 2, and the order with respect to Cl is 1