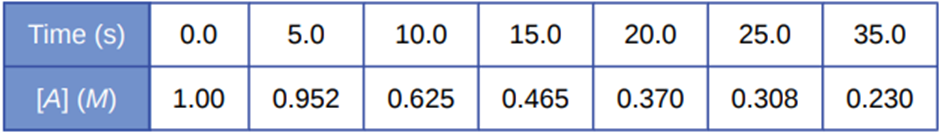
Chapter 12: Q26 E (page 704)
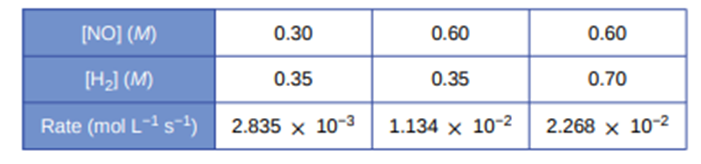
Hydrogen reacts with nitrogen monoxide to form dinitrogen monoxide (laughing gas) according to the equation:\({{\bf{H}}_{\bf{2}}}{\bf{(g) + 2NO(g)}} \to {{\bf{N}}_{\bf{2}}}{\bf{O(g) + }}{{\bf{H}}_{\bf{2}}}{\bf{O}}\).Determine the rate law, the rate constant, and the orders with respect to each reactant from the following data:
Short Answer
The rate law represented as\({\bf{rate = k(NO}}{{\bf{)}}^{\bf{2}}}{{\bf{(}}{{\bf{H}}_{\bf{2}}}{\bf{)}}^{\bf{1}}}\)
Reaction order with respect to NO is 2 and with respect to Cl is 1
Value of rate constant \({\bf{9}}{\bf{.0 \times 1}}{{\bf{0}}^{{\bf{ - 2}}}}{{\bf{L}}^{\bf{2}}}{\bf{Mo}}{{\bf{l}}^{{\bf{ - 2}}}}{\bf{s}}\)