Chapter 12: Q27 E (page 705)
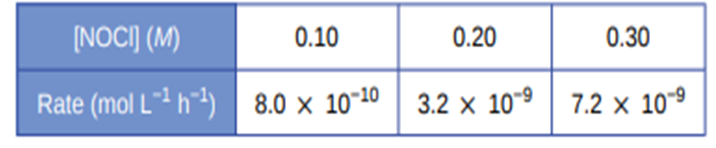
For the reaction\({\bf{A}} \to {\bf{B + C}}\), the following data were obtained at 30 °C:

- What is the order of the reaction with respect to (A), and what is the rate law?
- What is the rate constant?
Short Answer
- The order of reaction is 2 and the rate law is represented as \({\bf{Rate = k(A}}{{\bf{)}}^2}\)
- value of rate constant is \({\bf{7}}{\bf{.88 \times 1}}{{\bf{0}}^{{\bf{ - 3}}}}{{\bf{L}}^{\bf{1}}}{\bf{Mo}}{{\bf{l}}^{{\bf{ - 1}}}}{{\bf{s}}^{{\bf{ - 1}}}}\)