Chapter 12: Q23 E (page 704)
Nitrosyl chloride, NOCl, decomposes to NO and \({\bf{C}}{{\bf{l}}_{\bf{2}}}\).
\({\bf{2NOCl(g)}} \to {\bf{2NO(g) + C}}{{\bf{l}}_{\bf{2}}}{\bf{(g)}}\)
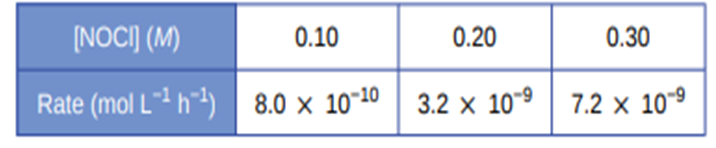
Determine the rate law, the rate constant, and the overall order for this reaction from the following data:
Short Answer
The rate law for decomposition of nitrosyl chloride is represented as
Rate of reaction= \({\bf{k(NOCl}}{{\bf{)}}^{\bf{2}}}\)
The overall order of the reaction is 2.
Value of rate constant is \({\bf{8 \times 1}}{{\bf{0}}^{{\bf{ - 8}}}}{\bf{mol}}{{\bf{L}}^{{\bf{ - 1}}}}{{\bf{h}}^{{\bf{ - 1}}}}\).