Chapter 12: Q22E (page 704)
Under certain conditions, the decomposition of ammonia on a metal surface gives the following data:
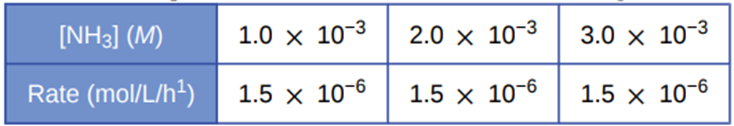
Determine the rate law, the rate constant, and the overall order for this reaction.
Short Answer
The rate law for decomposition of ammonia is equal to the rate constant. The value of rate law and rate constant is \({\bf{1}}{\bf{.5 \times 1}}{{\bf{0}}^{{\bf{ - 6}}}}{\bf{mol/L/h}}\)
The overall order of the reaction is zero.

