Chapter 12: Q12.8CYL (page 671)
If the initial concentration of butadiene is 0.0200 M, what is the concentration remaining after 20.0 min?
Short Answer
The concentration remaining after 20.0 min is 0.0196 mol/L.
Learning Materials
Features
Discover
Chapter 12: Q12.8CYL (page 671)
If the initial concentration of butadiene is 0.0200 M, what is the concentration remaining after 20.0 min?
The concentration remaining after 20.0 min is 0.0196 mol/L.
All the tools & learning materials you need for study success - in one app.
Get started for free
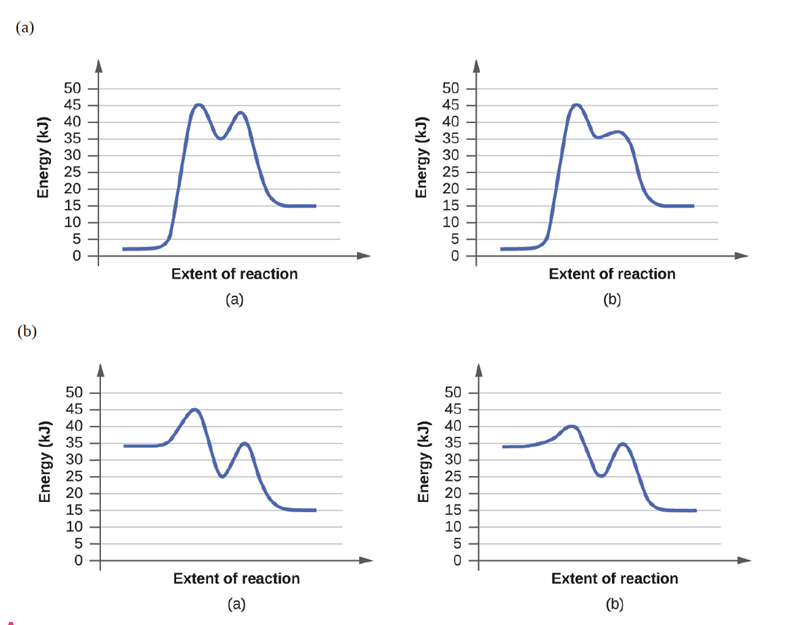
For each of the following pairs of reaction diagrams, identify which of the pairs iscatalyzed:
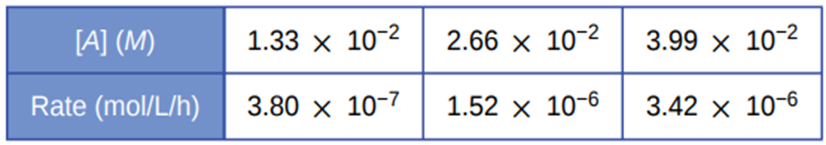
From the following data, determine the rate law, the rate constant, and the order with respect to A for the reaction \({\bf{A}} \to {\bf{2C}}\).
What is the difference between average rate, initial rate, and instantaneous rate?
Compare the functions of homogeneous and heterogeneous catalysts.
In an experiment, a sample of NaClO3 was 90% decomposed in 48 min. Approximately how long would this decomposition have taken if the sample had been heated 20°C higher?
What do you think about this solution?
We value your feedback to improve our textbook solutions.