Chapter 12: Q81E. (page 714)
For each of the following pairs of reaction diagrams, identify which of the pairs iscatalyzed:
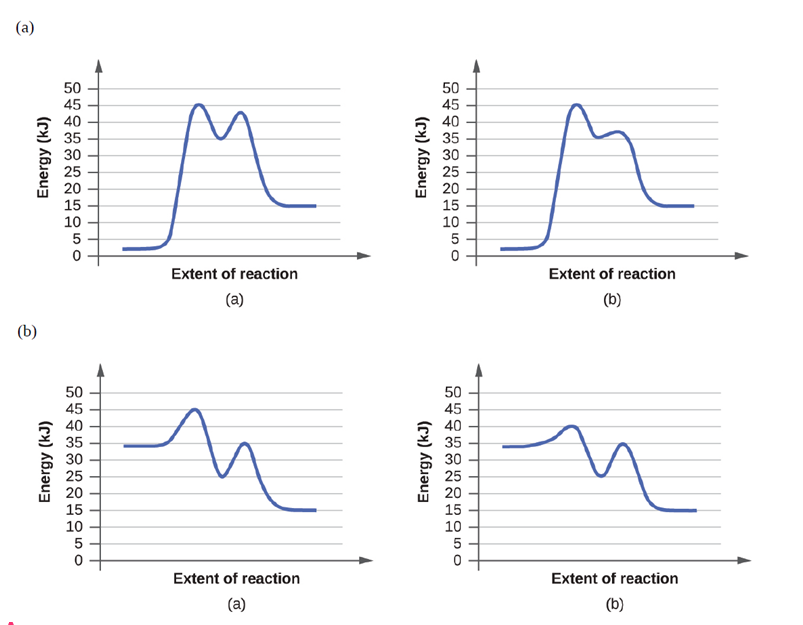
Short Answer
(a) Reaction diagram (b) is a catalyzed reaction.
(b) Reaction diagram (b) is a catalyzed reaction.
Learning Materials
Features
Discover
Chapter 12: Q81E. (page 714)
For each of the following pairs of reaction diagrams, identify which of the pairs iscatalyzed:

(a) Reaction diagram (b) is a catalyzed reaction.
(b) Reaction diagram (b) is a catalyzed reaction.
All the tools & learning materials you need for study success - in one app.
Get started for free
Atomic chlorine in the atmosphere reacts with ozone in the following pair of elementary reactions:
\({\bf{Cl + \;O3}}\left( {\bf{g}} \right){\bf{ - - - ClO}}\left( {\bf{g}} \right){\bf{ + \;O2}}\left( {\bf{g}} \right)\)(rate constant k1 )
\({\bf{ClO}}\left( {\bf{g}} \right){\bf{ + O - - - Cl}}\left( {\bf{g}} \right){\bf{ + \;O2}}\left( {\bf{g}} \right)\)(rate constant k2 )
Determine the overall reaction, write the rate law expression for each elementary reaction, identify any intermediates, and determine the overall rate law expression.
Doubling the concentration of a reactant increases the rate of a reaction four times. With this knowledge, answer the following questions:
The annual production of \({\bf{HN}}{{\bf{O}}_{\bf{3}}}\) in 2013 was 60 million metric tons Most of that was prepared by the following sequence of reactions, each run in a separate reaction vessel.
\(\begin{align}\left( a \right){\bf{ }}4N{H_3}{\bf{ }}\left( g \right){\bf{ }} + {\bf{ }}5{O_2}{\bf{ }}(g) \to 4NO\left( g \right){\bf{ }} + {\bf{ }}6{H_2}O\left( g \right)\\\left( b \right){\bf{ }}2NO\left( g \right){\bf{ }} + {\bf{ }}{O_{2{\bf{ }}}}(g) \to 2N{O_{2{\bf{ }}}}\left( g \right)\\\left( c \right){\bf{ }}3N{O_2}{\bf{ }}\left( g \right){\bf{ }} + {\bf{ }}{H_2}O(l) \to 2HN{O_3}(aq) + NO(g)\end{align}\)
The first reaction is run by burning ammonia in air over a platinum catalyst. This reaction is fast. The reaction in equation (c) is also fast. The second reaction limits the rate at which nitric acid can be prepared from ammonia. If equation (b) is second order in NO and first order in \({{\bf{O}}_{\bf{2}}}\), what is the rate of formation of \({\bf{N}}{{\bf{O}}_{\bf{2}}}\) when the oxygen concentration is 0.50 M and the nitric oxide concentration is 0.75 M? The rate constant for the reaction is \({\bf{5}}{\bf{.8 \times 1}}{{\bf{0}}^{{\bf{ - 6}}}}{\bf{ L}}{{\bf{ }}^{\bf{2}}}{\bf{ mo}}{{\bf{l}}^{{\bf{ - 2}}}}{\bf{ s}}{{\bf{ }}^{{\bf{ - 1}}}}\).
Experiments were conducted to study the rate of the reaction represented by this equation.(2)\({\rm{2NO(g) + 2}}{{\rm{H}}_{\rm{2}}}{\rm{(g) }} \to {\rm{ }}{{\rm{N}}_{\rm{2}}}{\rm{(g) + 2}}{{\rm{H}}_{\rm{2}}}{\rm{O(g)}}\)Initial concentrations and rates of reaction are given here.
| Experiment | Initial Concentration \(\left( {{\bf{NO}}} \right){\rm{ }}\left( {{\bf{mol}}/{\bf{L}}} \right)\) | Initial Concentration, \(\left( {{{\bf{H}}_{\bf{2}}}} \right){\rm{ }}\left( {{\bf{mol}}/{\bf{L}}} \right)\) | Initial Rate of Formation of \({{\bf{N}}_{\bf{2}}}{\rm{ }}\left( {{\bf{mol}}/{\bf{L}}{\rm{ }}{\bf{min}}} \right)\) |
| 1 | \({\bf{0}}.{\bf{0060}}\) | \({\bf{0}}.{\bf{00}}1{\bf{0}}\) | \({\bf{1}}.{\bf{8}} \times {\bf{1}}{{\bf{0}}^{ - {\bf{4}}}}\) |
| 2 | \({\bf{0}}.{\bf{0060}}\) | \({\bf{0}}.{\bf{00}}2{\bf{0}}\) | \({\bf{3}}.{\bf{6}} \times {\bf{1}}{{\bf{0}}^{ - {\bf{4}}}}\) |
| 3 | \({\bf{0}}.{\bf{00}}1{\bf{0}}\) | \({\bf{0}}.{\bf{0060}}\) | \({\bf{0}}.{\bf{30}} \times {\bf{1}}{{\bf{0}}^{ - {\bf{4}}}}\) |
| 4 | \({\bf{0}}.{\bf{00}}2{\bf{0}}\) | \({\bf{0}}.{\bf{0060}}\) | \({\bf{1}}.{\bf{2}} \times {\bf{1}}{{\bf{0}}^{ - {\bf{4}}}}\) |
Consider the following questions:(a) Determine the order for each of the reactants, \({\bf{NO}}\) and \({{\bf{H}}_{\bf{2}}}\), from the data given and show your reasoning.(b) Write the overall rate law for the reaction.(c) Calculate the value of the rate constant, k, for the reaction. Include units.(d) For experiment 2, calculate the concentration of \({\bf{NO}}\)remaining when exactly one-half of the original amount of \({{\bf{H}}_{\bf{2}}}\) had been consumed.(e) The following sequence of elementary steps is a proposed mechanism for the reaction.Step 1:Step 2:Step 3:Based on the data presented, which of these is the rate determining step? Show that the mechanism is consistent with the observed rate law for the reaction and the overall stoichiometry of the reaction.
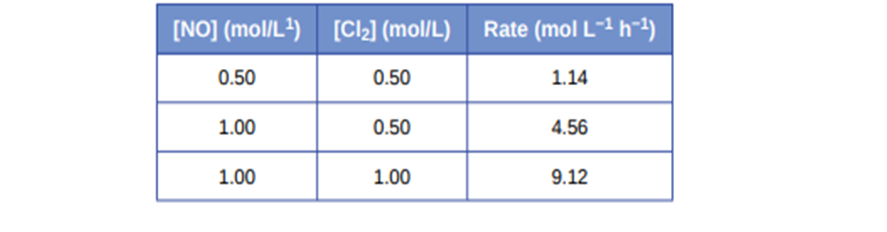
Nitrogen monoxide reacts with chlorine according to the equation:
2NO(g) + Cl\({}_2\)(g)⟶ 2NOCl(g) The following initial rates of reaction have been observed for certain reactant concentrations:
What is the rate law that describes the rate’s dependence on the concentrations of NO and Cl2? What is the rate constant? What are the orders with respect to each reactant?
What do you think about this solution?
We value your feedback to improve our textbook solutions.