Chapter 12: Q80E. (page 713)
For each of the following pairs of reaction diagrams, identify which of the pair iscatalyzed:

Short Answer
(a) Reaction diagram (b) is a catalyzed reaction.
(b) Reaction diagram (b) is a catalyzed reaction.
Learning Materials
Features
Discover
Chapter 12: Q80E. (page 713)
For each of the following pairs of reaction diagrams, identify which of the pair iscatalyzed:

(a) Reaction diagram (b) is a catalyzed reaction.
(b) Reaction diagram (b) is a catalyzed reaction.
All the tools & learning materials you need for study success - in one app.
Get started for free
Under certain conditions, the decomposition of ammonia on a metal surface gives the following data:
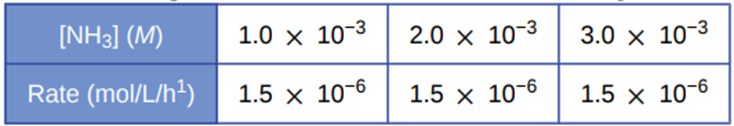
Determine the rate law, the rate constant, and the overall order for this reaction.
How much and in what direction will each of the following effect the rate of the reaction:
CO(g) + \({\bf{NO}}{}_{\bf{2}}\) (g)⟶ \({\bf{CO}}{}_{\bf{2}}\) (g) + NO(g) if the rate law for the reaction is rate =\({\bf{k(NO}}{}_{\bf{2}}{{\bf{)}}^{\bf{2}}}{\bf{a}}\)?
The annual production of \({\bf{HN}}{{\bf{O}}_{\bf{3}}}\) in 2013 was 60 million metric tons Most of that was prepared by the following sequence of reactions, each run in a separate reaction vessel.
\(\begin{align}\left( a \right){\bf{ }}4N{H_3}{\bf{ }}\left( g \right){\bf{ }} + {\bf{ }}5{O_2}{\bf{ }}(g) \to 4NO\left( g \right){\bf{ }} + {\bf{ }}6{H_2}O\left( g \right)\\\left( b \right){\bf{ }}2NO\left( g \right){\bf{ }} + {\bf{ }}{O_{2{\bf{ }}}}(g) \to 2N{O_{2{\bf{ }}}}\left( g \right)\\\left( c \right){\bf{ }}3N{O_2}{\bf{ }}\left( g \right){\bf{ }} + {\bf{ }}{H_2}O(l) \to 2HN{O_3}(aq) + NO(g)\end{align}\)
The first reaction is run by burning ammonia in air over a platinum catalyst. This reaction is fast. The reaction in equation (c) is also fast. The second reaction limits the rate at which nitric acid can be prepared from ammonia. If equation (b) is second order in NO and first order in \({{\bf{O}}_{\bf{2}}}\), what is the rate of formation of \({\bf{N}}{{\bf{O}}_{\bf{2}}}\) when the oxygen concentration is 0.50 M and the nitric oxide concentration is 0.75 M? The rate constant for the reaction is \({\bf{5}}{\bf{.8 \times 1}}{{\bf{0}}^{{\bf{ - 6}}}}{\bf{ L}}{{\bf{ }}^{\bf{2}}}{\bf{ mo}}{{\bf{l}}^{{\bf{ - 2}}}}{\bf{ s}}{{\bf{ }}^{{\bf{ - 1}}}}\).
The rate constant for the radioactive decay of 14C is \({\bf{1}}{\bf{.21 \times 1}}{{\bf{0}}^{{\bf{ - 4}}}}{\bf{ yea}}{{\bf{r}}^{{\bf{ - 1}}}}\). The products of the decay are nitrogen atoms and electrons (beta particles): \(\begin{aligned}{}_{\bf{6}}^{{\bf{14}}}{\bf{C}} \to _{\bf{6}}^{{\bf{14}}}{\bf{N + }}{{\bf{e}}^{\bf{ - }}}\\{\bf{rate = k(}}_{\bf{6}}^{{\bf{14}}}{\bf{C)}}\end{aligned}\).
What is the instantaneous rate of production of N atoms in a sample with a carbon-14 content of \({\bf{ 6}}{\bf{.5 \times 1}}{{\bf{0}}^{{\bf{ - 9 }}}}{\bf{M}}\)?
The element Co exists in two oxidation states, Co(II) and Co(III), and the ions form many complexes. The rate at which one of the complexes of Co(III) was reduced by Fe(II) in water was measured. Determine the activation energy of the reaction from the following data:
Temperature(K) | k(s-1) |
293 | 0.054 |
298 | 0.100 |
What do you think about this solution?
We value your feedback to improve our textbook solutions.