Chapter 12: Q1E (page 701)
What is the difference between average rate, initial rate, and instantaneous rate?
Short Answer
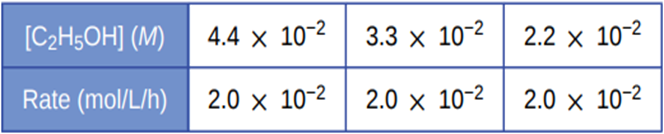
The main difference between the average rate, initial rate, and instantaneous rate is the amount of concentration and time taken. For average rate, change of concentration for a period is considered, whereas initial and instantaneous rates are for a particular time. The initial rate is the rate at the very beginning of the reaction, whereas instantaneous could be for any particular time at which the reaction is occurring.