Chapter 12: Q68 E (page 710)
Why are elementary reactions involving three or more reactants very uncommon?
Short Answer
The reaction rate depends on the number and orientation of collisions between the reactant molecules.
Learning Materials
Features
Discover
Chapter 12: Q68 E (page 710)
Why are elementary reactions involving three or more reactants very uncommon?
The reaction rate depends on the number and orientation of collisions between the reactant molecules.
All the tools & learning materials you need for study success - in one app.
Get started for free
Chemical reactions occur when reactants collide. What are two factors that may prevent a collision from producing a chemical reaction?
Based on the diagrams in Exercise 12.83, which of the reactions has the fastest rate? Which has the slowest rate?
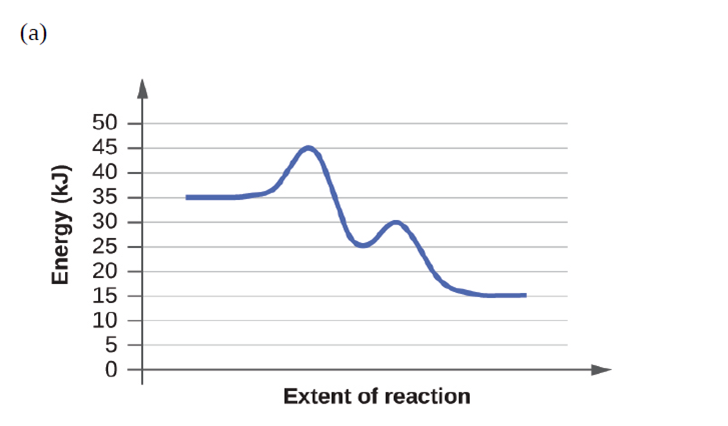
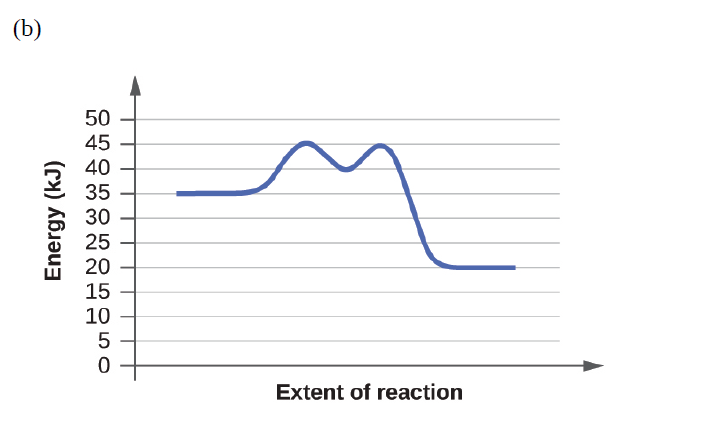
How much and in what direction will each of the following effect the rate of the reaction:
CO(g) + \({\bf{NO}}{}_{\bf{2}}\) (g)⟶ \({\bf{CO}}{}_{\bf{2}}\) (g) + NO(g) if the rate law for the reaction is rate =\({\bf{k(NO}}{}_{\bf{2}}{{\bf{)}}^{\bf{2}}}{\bf{a}}\)?
The first-order radioactive decay of iodine-131 exhibits a rate constant of 0.138 \({{\bf{d}}^{{\bf{ - 1}}}}\). What is the half-life for this decay?
In an experiment, a sample of NaClO3 was 90% decomposed in 48 min. Approximately how long would this decomposition have taken if the sample had been heated 20°C higher?
What do you think about this solution?
We value your feedback to improve our textbook solutions.