Chapter 12: Q51 E (page 708)
Chemical reactions occur when reactants collide. What are two factors that may prevent a collision from producing a chemical reaction?
Short Answer
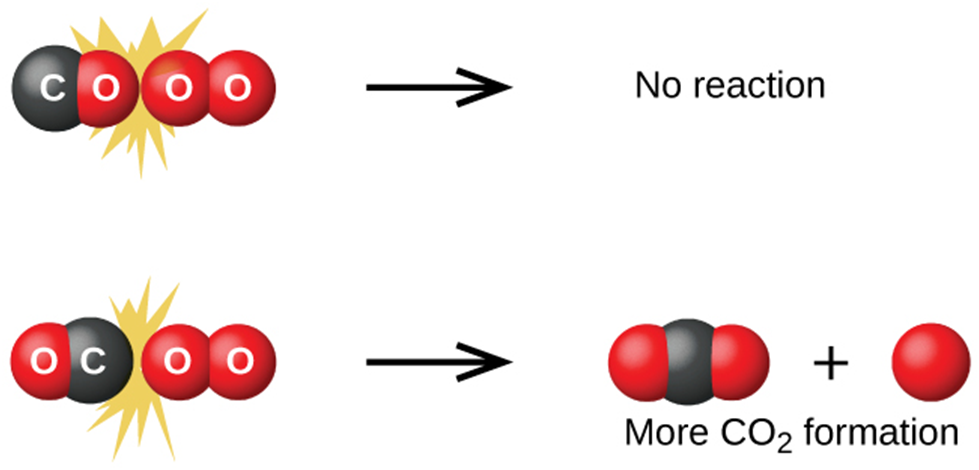
Collision theory is a theory that is used to predict the rates of chemical reactions, particularly those involving gases. The collision theory holds that for a reaction to take place, the reacting species (atoms or molecules) must come into contact with one another.
Collision theory is founded on the following premises:
The reacting species must collide in such a way that contact between the atoms that will become bonded together in the product is possible.
The collision must have enough energy to allow mutual penetration of the valence of the reacting species. Shells, allowing electrons to rearrange and form new bonds (and new chemical species).