Chapter 12: Q12.5CYL (page 666)
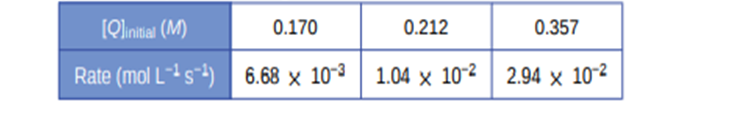
Use the provided initial rate data to derive the rate law for the reaction whose equation is: \({\bf{OC}}{{\bf{l}}^ - }\)(aq) + \({{\bf{I}}^ - }\)(aq) ⟶OI−(aq) +\({\bf{C}}{{\bf{l}}^ - }\)(aq)
Trial | (\({\bf{OC}}{{\bf{l}}^ - }\)) (mol/L) | (\({{\bf{I}}^ - }\)) (mol/L) | Initial Rate (mol/L/s) |
1. | 0.0040 | 0.0020 | 0.00184 |
2. | 0.0020 | 0.0040 | 0.00092 |
3. | 0.0020 | 0.0020 | 0.00046 |
Determine the rate law expression and the value of the rate constant k with appropriate units for this reaction.
Short Answer
The rate, \(r = k\left( {{\bf{OC}}{{\bf{l}}^ - }} \right)\left( {{{\bf{l}}^ - }} \right)\) with rate constant k = 5.75 × 104mol-2L2s-1.