Chapter 12: Q85E. (page 716)
Based on the diagrams in Exercise 12.83, which of the reactions has the fastest rate? Which has the slowest rate?
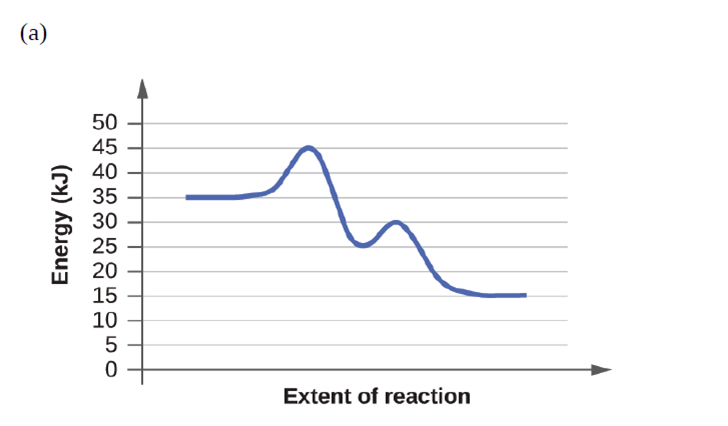
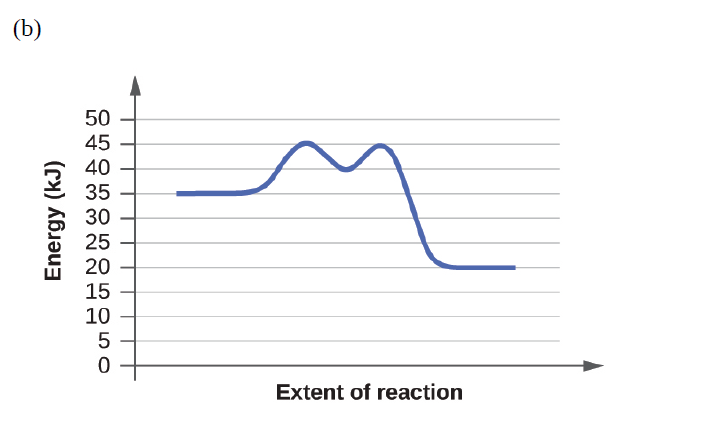
Short Answer
Reactions (a) and (b) have the same rate.
Learning Materials
Features
Discover
Chapter 12: Q85E. (page 716)
Based on the diagrams in Exercise 12.83, which of the reactions has the fastest rate? Which has the slowest rate?


Reactions (a) and (b) have the same rate.
All the tools & learning materials you need for study success - in one app.
Get started for free
Use the provided initial rate data to derive the rate law for the reaction whose equation is: \({\bf{OC}}{{\bf{l}}^ - }\)(aq) + \({{\bf{I}}^ - }\)(aq) ⟶OI−(aq) +\({\bf{C}}{{\bf{l}}^ - }\)(aq)
Trial | (\({\bf{OC}}{{\bf{l}}^ - }\)) (mol/L) | (\({{\bf{I}}^ - }\)) (mol/L) | Initial Rate (mol/L/s) |
1. | 0.0040 | 0.0020 | 0.00184 |
2. | 0.0020 | 0.0040 | 0.00092 |
3. | 0.0020 | 0.0020 | 0.00046 |
Determine the rate law expression and the value of the rate constant k with appropriate units for this reaction.
Describe the effect of each of the following on the rate of the reaction of magnesium metal with a solution of hydrochloric acid: the molarity of the hydrochloric acid, the temperature of the solution, and the size of the pieces of magnesium.
Alcohol is removed from the bloodstream by a series of metabolic reactions. The first reaction produces acetaldehyde; then other products are formed. The following data have been determined for the rate at which alcohol is removed from the blood of an average male, although individual rates can vary by 25–30%. Women metabolize alcohol a little more slowly than men:
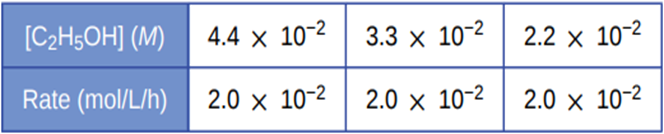
Determine the rate equation, the rate constant, and the overall order for this reaction.
How do the rate of a reaction and its rate constant differ?
Fluorine-18 is a radioactive isotope that decays by positron emission to form oxygen-18 with a half-life of 109.7 min. (A positron is a particle with the mass of an electron and a single unit of positive charge; the equation is (\({_{{\bf{518}}}^{\bf{9}}}{\bf{F}}\)⟶\({_{{\bf{18}}}^{\bf{8}}}{\bf{O + e - }}\).) Physicians use\(^{{\bf{18}}}{\bf{F}}\)to study the brain by injecting a quantity of fluoro-substituted glucose into the blood of a patient. The glucose accumulates in the regions where the brain is active and needs nourishment.
(a) What is the rate constant for the decomposition of fluorine-18?
(b) If a sample of glucose containing radioactive fluorine-18 is injected into the blood, what percent of the radioactivity will remain after 5.59 h?
(c) How long does it take for 99.99% of the\(^{{\bf{18}}}{\bf{F}}\)to decay?
What do you think about this solution?
We value your feedback to improve our textbook solutions.