Chapter 12: Q12E (page 702)
How do the rate of a reaction and its rate constant differ?
Short Answer
The rate of a reaction is proportional to its rate constant, and it typically increases with the concentration of reactants.
Learning Materials
Features
Discover
Chapter 12: Q12E (page 702)
How do the rate of a reaction and its rate constant differ?
The rate of a reaction is proportional to its rate constant, and it typically increases with the concentration of reactants.
All the tools & learning materials you need for study success - in one app.
Get started for free
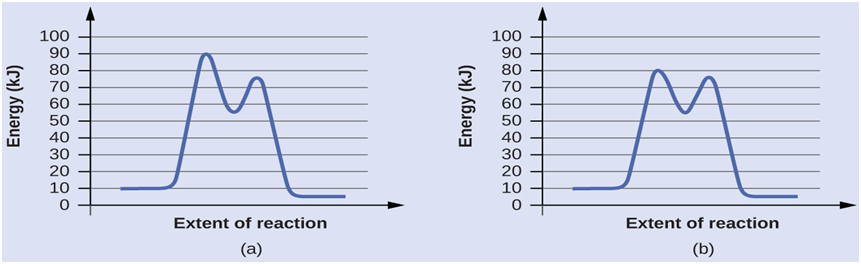
Determine which of the two diagrams here (both for the same reaction) involves a catalyst, and identify the activation energy for the catalyzed reaction:
(a) Multiply 2.334 cm and 0.320 cm.(b) Divide 55.8752 m by 56.53 s.
Acetaldehyde decomposes when heated to yield methane and carbon monoxide according to the equation: \({\bf{C}}{{\bf{H}}_{\bf{3}}}{\bf{CHO}}\)(g) ⟶\({\bf{C}}{{\bf{H}}_{\bf{4}}}\)(g) +\({\bf{CO}}\)(g)
Determine the rate law and the rate constant for the reaction from the following experimental data:
Trial | (\({\bf{C}}{{\bf{H}}_{\bf{3}}}{\bf{CHO}}\)) (mol/L) | \(\frac{{ - \Delta \left( {{\bf{C}}{{\bf{H}}_{\bf{3}}}{\bf{CHO}}} \right)}}{{\Delta t}}\)(mol )(Ls−1) |
1. | 1.75 × 10−3 | 2.06 × 10−11 |
2. | 3.50 × 10−3 | 8.24 × 10−11 |
3. | 7.00 × 10−3 | 3.30 × 10−10 |
In a transesterification reaction, a triglyceride reacts with an alcohol to form an ester and glycerol. Many students learn about the reaction between methanol (\({\bf{C}}{{\bf{H}}_{\bf{3}}}{\bf{OH}}\)) and ethyl acetate (\({\bf{C}}{{\bf{H}}_{\bf{3}}}{\bf{C}}{{\bf{H}}_{\bf{2}}}{\bf{OCOC}}{{\bf{H}}_{\bf{3}}}\)) as a sample reaction before studying the chemical reactions that produce biodiesel:
\({\bf{C}}{{\bf{H}}_{\bf{3}}}{\bf{OH + C}}{{\bf{H}}_{\bf{3}}}{\bf{C}}{{\bf{H}}_{\bf{2}}}{\bf{OCOC}}{{\bf{H}}_{\bf{3}}}{\bf{ - - - C}}{{\bf{H}}_{\bf{3}}}{\bf{OCOC}}{{\bf{H}}_{\bf{3}}}{\bf{ + C}}{{\bf{H}}_{\bf{3}}}{\bf{C}}{{\bf{H}}_{\bf{2}}}{\bf{OH}}\).The rate law for the reaction between methanol and ethyl acetate is, under certain conditions, determined to be: rate =\(k\left( {{\bf{C}}{{\bf{H}}_{\bf{3}}}{\bf{OH }}} \right)\). What is the order of reaction with respect to methanol and ethyl acetate, and what is the overall order of reaction?
For the reaction\({\bf{A}} \to {\bf{B + C}}\), the following data were obtained at 30 °C:

What do you think about this solution?
We value your feedback to improve our textbook solutions.