Chapter 19: Q. 9 (page 543)
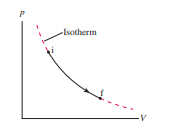
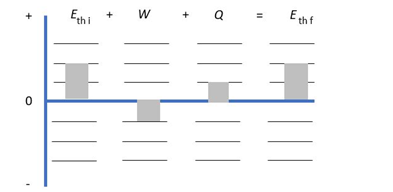
Draw a first-law bar chart (see Figure 19.12) for the gas process in Figure EX19.9
Short Answer
The work done and heat is same magnitude but heat is positive and work done is negative
Learning Materials
Features
Discover
Chapter 19: Q. 9 (page 543)
Draw a first-law bar chart (see Figure 19.12) for the gas process in Figure EX19.9

The work done and heat is same magnitude but heat is positive and work done is negative

All the tools & learning materials you need for study success - in one app.
Get started for free
Radiation from the head is a major source of heat loss from the human body. Model a head as a 20-cm-diameter, 20-cm-tall cylinder with a flat top. If the body’s surface temperature is 35°C, what is the net rate of heat loss on a chilly 5°C day? All skin, regardless of color, is effectively black in the infrared where the radiation occurs, so use an emissivity of 0.95.
512 g of an unknown metal at a temperature of 15°C is dropped into a 100 g aluminum container holding 325 g of water at 98°C. A short time later, the container of water and metal stabilizes at a new temperature of 78°C. Identify the metal.
of gas are at and a pressure of . How much work must be done on the gas to compress it to one-third of its initial volume at (a) constant temperature and (b) constant pressure? (c) Show both processes on a singlediagram.
A gas cylinder holds 0.10 mol of O2 at 150C and a pressure
of 3.0 atm. The gas expands adiabatically until the volume is doubled.
What are the final (a) pressure and (b) temperature?
A container holds 1.0 g of oxygen at a pressure of 8.0 atm.
a. How much heat is required to increase the temperature by 100°C at constant pressure?
b. How much will the temperature increase if this amount of heat energy is transferred to the gas at constant volume?
What do you think about this solution?
We value your feedback to improve our textbook solutions.