Chapter 19: Q. 49 (page 545)
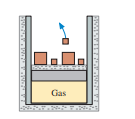
of gas are at and a pressure of . How much work must be done on the gas to compress it to one-third of its initial volume at (a) constant temperature and (b) constant pressure? (c) Show both processes on a singlediagram.
Short Answer
a) Amount of work must be done on the gas to compress it to one-third of its initial volume at constant temperature is
b) Amount of work must be done on the gas to compress it to one-third of its initial volume at constant pressure is
c) The diagrammatic representation of diagram is