Chapter 19: Q. 65 (page 546)
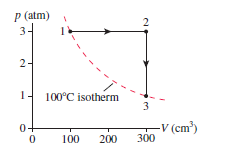
of nitrogen gas at STP are pressurized in an isochoric process to a pressure of . What are (a) the final temperature, (b) the work done on the gas, (c) the heat transfer to the gas, and (d) the pressure ratio ? (e) Show the process on a diagram, using proper scales on both axes.
Short Answer
a) The final temperature is
b) The work done on the gas is Zero.
c) The heat transfer to the gas is
d) The pressure ratio is or
e) The diagrammatic representation is: