Chapter 19: Q. 60 (page 546)
Your laboratory assignment for the week is to measure the specific heat ratio of carbon dioxide. The gas is contained in a cylinder with a movable piston and a thermometer. When the piston is withdrawn as far as possible, the cylinder's length is . You decide to push the piston in very rapidly by various amounts and, for each push, to measure the temperature of the carbon dioxide. Before each push, you withdraw the piston all the way and wait several minutes for the gas to come to the room temperature of . Your data are as follows:
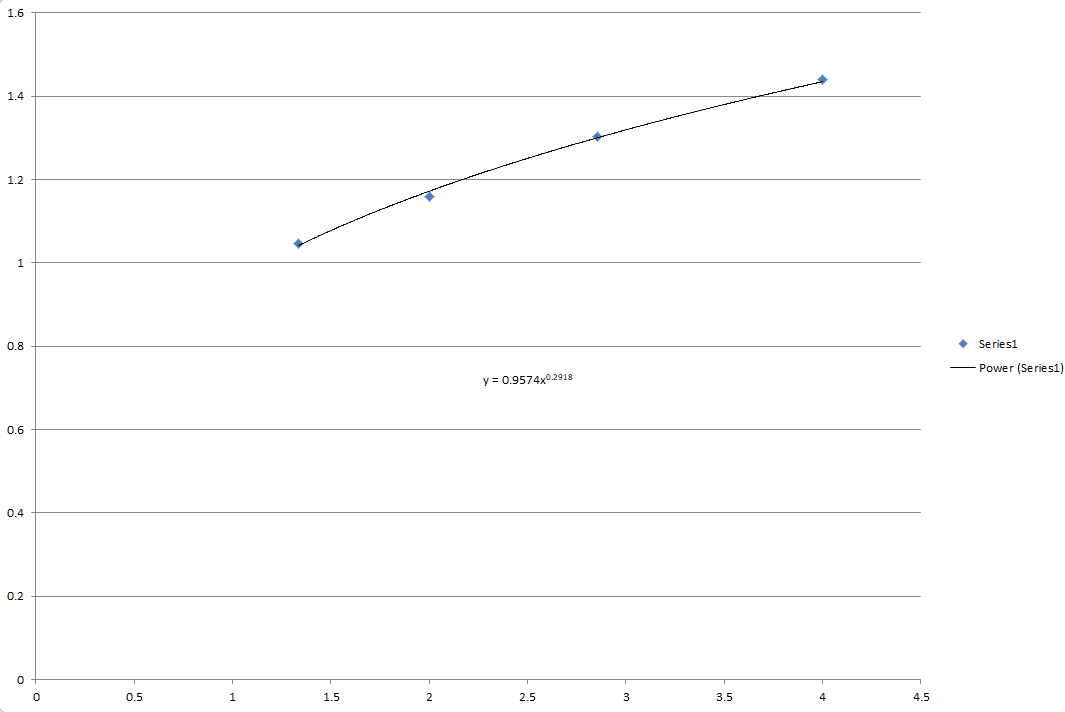
Use the best-fit line of an appropriate graph to determine for carbon dioxide.
Short Answer
The best-fit line of an appropriate graph to determine for carbon dioxide is