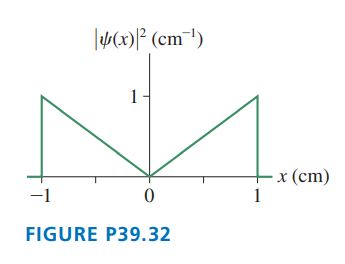Chapter 39: Q.13. (page 1137)
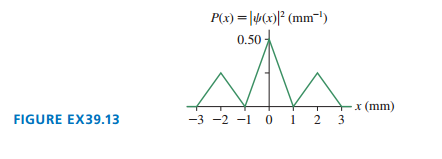
FIGURE EX39.13 shows the probability density for an electron that has passed through an experimental apparatus. What is the probability that the electron will land in a -wide strip at (a) , (b), (c) , and (d) ?
Short Answer
a. The probability of electron will land in a wide string at a position of is .
b. The probability of electron will land in a wide string at a position of is .
c. The probability of electron will land in a wide string at a position of is 0.
d. The probability of electron will land in a wide string at a position of is .