Chapter 39: Q. 41 (page 1118)
What is the smallest one-dimensional box in which you can confine an electron if you want to know for certain that the electron's speed is no more than ?
Short Answer
The smallest one-dimensional box is
Learning Materials
Features
Discover
Chapter 39: Q. 41 (page 1118)
What is the smallest one-dimensional box in which you can confine an electron if you want to know for certain that the electron's speed is no more than ?
The smallest one-dimensional box is
All the tools & learning materials you need for study success - in one app.
Get started for free
Consider a single-slit diffraction experiment using electrons. Using Figure 39.5 as a model, draw
a. A dot picture showing the arrival positions of the first or electrons.
b. A graph of for the electrons on the detection screen.
c. A graph of for the electrons. Keep in mind that , as a wave-like function, oscillates between positive and negative.
Andrea, whose mass is , thinks she's sitting at rest in her 5.0-m-long dorm room as she does her physics homework. Can Andrea be sure she's at rest? If not, within what range is her velocity likely to be?
The probability density for finding a particle at position x is P1x2 = • a 11 - x2 -1 mm … x 6 0 mm b11 - x2 0 mm … x … 1 mm and zero elsewhere. a. You will learn in Chapter 40 that the wave function must be a continuous function. Assuming that to be the case, what can you conclude about the relationship between a and b? b. Determine values for a and b. c. Draw a graph of the probability density over the interval -2 mm … x … 2 mm. d. What is the probability that the particle will be found to the left of the origin?
In an interference experiment with electrons, you find the most intense fringe is at x = 7.0 cm. There are slightly weaker fringes at x= 6.0 and 8.0 cm, still weaker fringes at x = 4.0 and 10.0 cm, and two very weak fringes at x= 1.0 and 13.0 cm. No electrons are detected at x <0 cm or x> 14 cm.
a. Sketch a graph of is for these electrons.
b. Sketch a possible graph of .
c. Are there other possible graph for? If so draw one
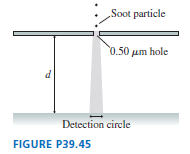
Soot particles, from incomplete combustion in diesel engines, are typically in diameter and have a density of . FIGURE P39.45 shows soot particles released from rest, in vacuum, just above a thin plate with a -diameter holeroughly the wavelength of visible light. After passing through the hole, the particles fall distance d and land on a detector. If soot particles were purely classical, they would fall straight down and, ideally, all land in a -diameter circle. Allowing for some experimental imperfections, any quantum effects would be noticeable if the circle diameter were . How far would the particles have to fall to fill a circle of this diameter?
What do you think about this solution?
We value your feedback to improve our textbook solutions.