Chapter 38: Q. 53 (page 1116)
An electron confined in a one-dimensional box emits a 200 nm photon in a quantum jump from n=2 to n =1. What is the length of the box?
Short Answer
Tthe length of the box is
Learning Materials
Features
Discover
Chapter 38: Q. 53 (page 1116)
An electron confined in a one-dimensional box emits a 200 nm photon in a quantum jump from n=2 to n =1. What is the length of the box?
Tthe length of the box is
All the tools & learning materials you need for study success - in one app.
Get started for free
In a photoelectric-effect experiment, the stopping potential was measured for several different wavelengths of incident light. The data are as follows:
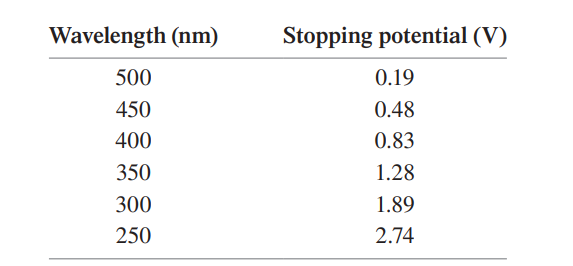
Use an appropriate graph of the data to determine
(a) the metal used for the cathode and
(b) an experimental value for Planck’s constant .
How many photoelectrons are ejected per second in the experiment represented by the graph of FIGURE EX38.2?
How much energy does it take to ionize a hydrogen atom that is in its first excited state?
Consider a hydrogen atom in stationary state n.
a. Show that the orbital period of an electron in quantum state n is, and find a numerical value for
b. On average, an atom stays in the n = 2 state for 1.6 ns before undergoing a quantum jump to the n = 1 state. On average, how many revolutions does the electron make before the quantum jump?
What lie the winding of the Irasuliens? , and in the hydronics ? In what spectral range do these lie?
What do you think about this solution?
We value your feedback to improve our textbook solutions.