Chapter 38: Q. 52 (page 1116)
A proton emits a gamma-ray photon with energy 2.0 MeV in a quantum jump from n =2 to n= 1.
Short Answer
The length of the box is:
Learning Materials
Features
Discover
Chapter 38: Q. 52 (page 1116)
A proton emits a gamma-ray photon with energy 2.0 MeV in a quantum jump from n =2 to n= 1.
The length of the box is:
All the tools & learning materials you need for study success - in one app.
Get started for free
What is the radius of a hydrogen atom whose electron moves at ?
A 100 W incandescent lightbulb emits about 5 W of visible light. (The other 95 W are emitted as infrared radiation or lost as heat to the surroundings.) The average wavelength of the visible light is about 600 nm, so make the simplifying assumption that all the light has this wavelength. How many visible-light photons does the bulb emit per second?
The diameter of the nucleus is about . A simple model of the nucleus is that protons and neutrons are confined within a one-dimensional box of length . What are the first three energy levels, in , for a proton in such a box?
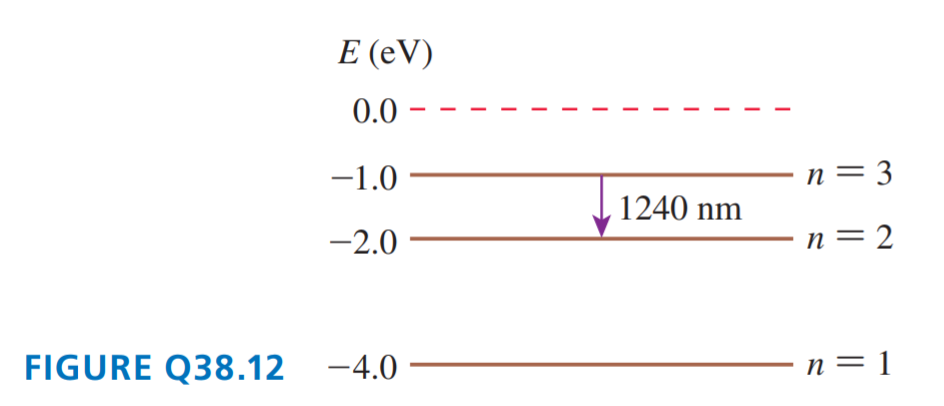
FIGURE Q38.12 shows the energy-level diagram of Element X.
a. What is the ionization energy of Element X?
b. An atom in the ground state absorbs a photon, then emits a photon with a wavelength of 1240 nm. What was the energy of the photon that was absorbed?
c. An atom in the ground state has a collision with an electron, then emits a photon with a wavelength of 1240 nm. What conclusion can you draw about the initial kinetic energy of the electron?
The wavelengths of light emitted by a firefly span the visible spectrum but have maximum intensity near 550 nm. A typical flash lasts for 100 ms and has a power output of 1.2 mW. How many photons does a firefly emit in one flash if we assume that all light is emitted at the peak intensity wavelength of 550 nm?
What do you think about this solution?
We value your feedback to improve our textbook solutions.