Chapter 38: Q. 29 (page 1115)
What is the radius of a hydrogen atom whose electron moves at ?
Short Answer
The radius of the hydrogen atom
Learning Materials
Features
Discover
Chapter 38: Q. 29 (page 1115)
What is the radius of a hydrogen atom whose electron moves at ?
The radius of the hydrogen atom
All the tools & learning materials you need for study success - in one app.
Get started for free
Consider a hydrogen atom in stationary state n.
a. Show that the orbital period of an electron in quantum state n is, and find a numerical value for
b. On average, an atom stays in the n = 2 state for 1.6 ns before undergoing a quantum jump to the n = 1 state. On average, how many revolutions does the electron make before the quantum jump?
The absorption spectrum of an atom consists of the wavelengths 200 nm, 300 nm, and 500 nm. (a) Draw the atom’s energy-level diagram. (b) What wavelengths are seen in the atom’s emission spectrum?
A beam of electrons is incident upon a gas of hydrogen atoms.
a. What minimum speed must the electrons have to cause the emission of 686 nm light from thetransition of hydrogen?
b. Through what potential difference must the electrons be accelerated to have this speed?
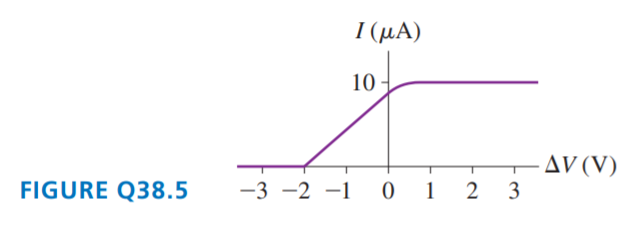
FIGURE Q38.5 is the current-versus-potential-difference graph for a photoelectric-effect experiment with an unknown metal. If classical physics provided the correct description of the photoelectric effect, how would the graph look if:
a. The light was replaced by an equally intense light with a shorter wavelength? Draw it.
b. The metal was replaced by a different metal with a smaller work function? Draw it.
Consider a hydrogen atom in stationary state n. (a) Show that the orbital period of an electron in quantum state n is and find a numerical value for (b) On average, an atom stays in the n = 2 state for 1.6 ns before undergoing a quantum jump to the n = 1 state. On average, how many revolutions does the electron make before the quantum jump?
What do you think about this solution?
We value your feedback to improve our textbook solutions.