Chapter 38: Q. 2 (page 1114)
How many photoelectrons are ejected per second in the experiment represented by the graph of FIGURE EX38.2?
Short Answer
There arephoto electrons are ejected per seconds.
Learning Materials
Features
Discover
Chapter 38: Q. 2 (page 1114)
How many photoelectrons are ejected per second in the experiment represented by the graph of FIGURE EX38.2?
There arephoto electrons are ejected per seconds.
All the tools & learning materials you need for study success - in one app.
Get started for free
Compton scattering is relevant not only to -ray photons but, even more so, to higher energy gamma-ray photons. Suppose a gamma-ray photon backscatter (i.e., is scattered back toward the source) from a free electron. Afterward, what is the electron’s velocity in ?
An FM radio station broadcasts with a power of 10 kW at a frequency of 101 MHz.
a. How many photons does the antenna emit each second?
b. Should the broadcast be treated as an electromagnetic wave or discrete photons? Explain.
In a photoelectric-effect experiment, the stopping potential was measured for several different wavelengths of incident light. The data are as follows:
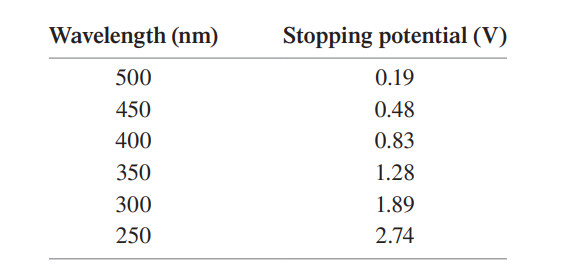
Use an appropriate graph of the data to determine
(a) the metal used for the cathode and
(b) an experimental value for Planck’s constant .
20.What is the quantum number of an electron confined in a 3.0-nm-long one-dimensional box if the electron's de Broglie wavelength is ?
I The allowed energies of a simple atom are
a. Draw the atom's energy-level diagram. Label each level with the energy and the quantum number.
b. What wavelengths appear in the atom's emission spectrum?
c. What wavelengths appear in the atom's absorption spectrum?
What do you think about this solution?
We value your feedback to improve our textbook solutions.