Chapter 40: One-Dimensional Quantum Mechanics
Q. 26
Suppose that and are both solutions to the Schrödinger equation for the same potential energy . Prove that the superposition is also a solution to the Schrödinger equation.
Q. 27
A diameter water droplet is moving with a speed of in a long box.
a. Estimate the particle’s quantum number.
b. Use the correspondence principle to determine whether quantum mechanics is needed to understand the particle’s motion or if it is “safe” to use classical physics.
Q. 28
Figure 40.27a modeled a hydrogen atom as a finite potential well with rectangular edges. A more realistic model of a hydrogen atom, although still a one-dimensional model, would be the electron + proton electrostatic potential energy in one dimension:
a. Draw a graph of U(x) versus x. Center your graph at .
b. Despite the divergence at , the Schrödinger equation can be solved to find energy levels and wave functions for the electron in this potential. Draw a horizontal line across your graph of part a about one-third of the way from the bottom to the top. Label this line , then, on this line, sketch a plausible graph of the wave function.
c. Redraw your graph of part a and add a horizontal line about two-thirds of the way from the bottom to the top. Label this line , then, on this line, sketch a plausible graph of the wave function.
Q. 29
Model an atom as an electron in a rigid box of length , roughly twice the Bohr radius.
a. What are the four lowest energy levels of the electron?
b. Calculate all the wavelengths that would be seen in the emission spectrum of this atom due to quantum jumps between these four energy levels. Give each wavelength a label to indicate the transition.
c. Are these wavelengths in the infrared, visible, or ultraviolet portion of the spectrum?
d. The stationary states of the Bohr hydrogen atom have negative energies. The stationary states of this model of the atom have positive energies. Is this a physically significant difference? Explain.
e. Compare this model of an atom to the Bohr hydrogen atom. In what ways are the two models similar? Other than the signs of the energy levels, in what ways are they different?
Q. 3
A particle in a potential well is in the quantum state. How many peaks are in the probability density ?
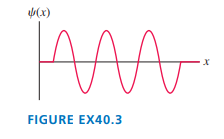
Q. 3
| FIGURE EX shows the wave function of an electron in a rigid box. The electron energy is. How long is the box?
Q. 30
a. Derive an expression for , the wavelength of light emitted by a particle in a rigid box during a quantum jump from
b. In what length rigid box will an electron undergoing a transition emit light with a wavelength of ? This is the wavelength of a ruby laser
Q. 31
Show that the normalization constant for the wave functions of a particle in a rigid box has the value given in Equation 40.26.
Q. 32
A particle confined in a rigid one-dimensional box of length has an energy level and an adjacent energy level .
a. Determine the values of n and n + 1.
b. Draw an energy-level diagram showing all energy levels from 1 through n + 1. Label each level and write the energy beside it.
c. Sketch the n + 1 wave function on the n + 1 energy level.
d. What is the wavelength of a photon emitted in the transition? Compare this to a typical visible-light wavelength.
e. What is the mass of the particle? Can you identify it?
Q. 33
Consider a particle in a rigid box of length L. For each of the states and :
a. Sketch graphs of . Label the points and .
b. Where, in terms of L, are the positions at which the particle is most likely to be found?
c. Where, in terms of L, are the positions at which the particle is least likely to be found?
d. Determine, by examining your graphs, if the probability of finding the particle in the left one-third of the box is less than, equal to, or greater than . Explain your reasoning.
e. Calculate the probability that the particle will be found in the left one-third of the box
