Chapter 0: Q13P (page 1)
Titration of EDTA with metal ion. Use Equation 12-12 to reproduce the results of Exercise 12-C
Short Answer
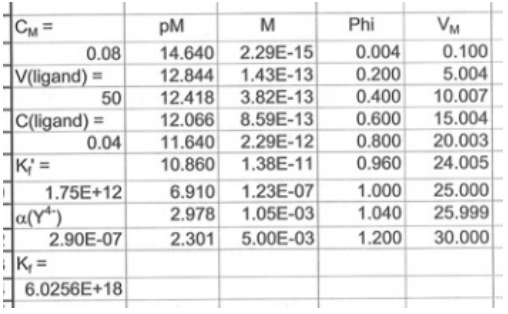
The spreadsheet developed for the titration reaction is shown below is shown below
Learning Materials
Features
Discover
Chapter 0: Q13P (page 1)
Titration of EDTA with metal ion. Use Equation 12-12 to reproduce the results of Exercise 12-C
The spreadsheet developed for the titration reaction is shown below is shown below

All the tools & learning materials you need for study success - in one app.
Get started for free
25-1 What should be the volume flow rate and injected volume for a 1.5-mm-diameter column?
Which will be more soluble (moles of metal dissolved per litre of solution), ? Give an example of a chemical reaction that might occur that would reverse the predicted solubilities.
(a) What is the pH at the equivalence point when 0.030 0 M NaF is titrated with 0.060 0 M HClO4?
(b) Why would an indicator endpoint probably not be useful in this titration?
Calculate the isoelectric and isoionicpHof 0.010Mthreonine.
What mass of protein gives a corrected absorbance of ?
What do you think about this solution?
We value your feedback to improve our textbook solutions.