Chapter 0: Q13P (page 1)
How many milliliters of 0.202MNaOHshould be added to 25.0 mlof 0.0233Msalicylic acid (2-hydroxybenzoic acid) to adjust the pHto 3.50?
Short Answer
The required volume of
Learning Materials
Features
Discover
Chapter 0: Q13P (page 1)
How many milliliters of 0.202MNaOHshould be added to 25.0 mlof 0.0233Msalicylic acid (2-hydroxybenzoic acid) to adjust the pHto 3.50?
The required volume of
All the tools & learning materials you need for study success - in one app.
Get started for free
Write the reaction of phosphoric acid and the reaction of disodium oxalate.
The efficiency of a gas chromatography column is measured by a parameter called plate height (H, m m) , which is related to the gas flow rate (u, mL/min) by the van Deemter equation: H = A + B / u + Cu , where A , B , and C are constants. Prepare a spreadsheet with a graph showing values of as a function of u for u =4,6,8,10,20,20,40,50,60,70,80,90 and 10 mL/min. Use the values A = 1.65 m m , B = 25.8 m m .m L/min, and C = 0.023 6 m m.min/mL.
Protein and carbohydrates provide, whereas fat gives . (Remember that 1 Calorie , with a capital C, is really .) The weight percentages of these components in some foods are
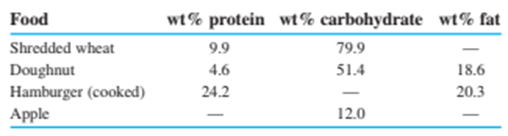
Calculate the number of calories per gram and calories per ounce in each of these foods. (Use Table 1-4to convert grams into ounces, remembering that there are 16ounces in 1pound.)
Find the minimum detectable concentration if the average of the blanks is and .
The iodide content of a commercial mineral water was measured by two methods that produced wildly different results. Method A found milligrams of per literand method B found 0.009mg/L. When was added to the water, the content found by method A increased each time that morewas added, but results from method B were unchanged. Which of the Terms to Understand describes what is occurring in these measurements? Explain your answer. Which result is more reliable?
What do you think about this solution?
We value your feedback to improve our textbook solutions.