Chapter 0: Q5-2 TY (page 1)
Find the minimum detectable concentration if the average of the blanks is and .
Short Answer
The minimum detectable concentration was calculated as .
Learning Materials
Features
Discover
Chapter 0: Q5-2 TY (page 1)
Find the minimum detectable concentration if the average of the blanks is and .
The minimum detectable concentration was calculated as .
All the tools & learning materials you need for study success - in one app.
Get started for free
The relative retention for two compounds in gas chromatography is 1.068 on a column with a plate height of 0.520mm.The retention factor for compound 1 is 5.16.
(a) Find the retention factor for compound 2.
(b) What length of column will separate the compounds with a resolution of 1.50 ?
(c) The retention time for air () is 2.00 min. If the number of plates is the same for both compounds, Findfor each peak.
(d) If the volume ratio of stationary phase to mobile phase is 0.30,Find the partition coefficient for compound 1.
How many milliliters of 2.15% alcoholic dimethylglyoxime should be used to provide a 50.0% excess for Reaction 27-7 with 0.998 4 g of steel containing 2.07 wt% Ni? Assume that the density of the dimethylglyoxime solution is 0.790 g/mL.
Find the area from tohours.
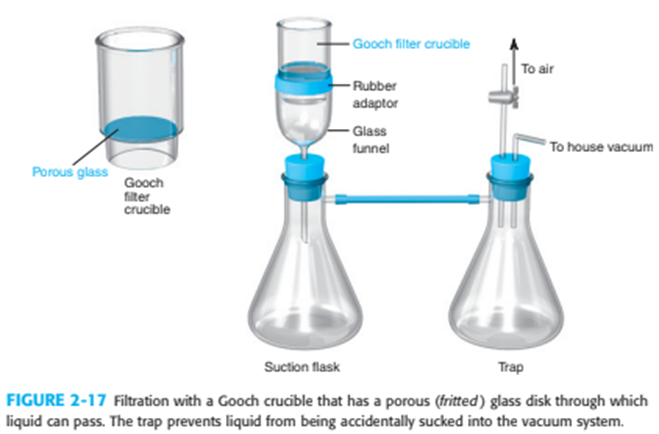
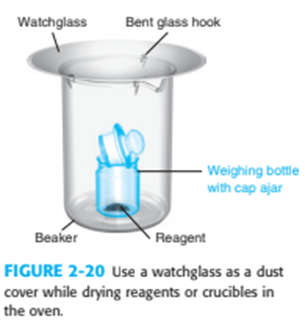
What is the purpose of the trap in Figure 2-17 and the watch glass in Figure 2-20?
Effect of concentration in the titration of weak acid with strong base.Use your spreadsheet from Problem 11-66 to prepare a family of titration curves for pKa = 6, with the following combinations of concentrations: (a) Ca = 20 mM, Cb = 100 mM; (b) Ca = 2 mM, Cb = 10 mM; (c) Ca = 0.2 mM, Cb = 1 mM.
What do you think about this solution?
We value your feedback to improve our textbook solutions.